Ligand binding
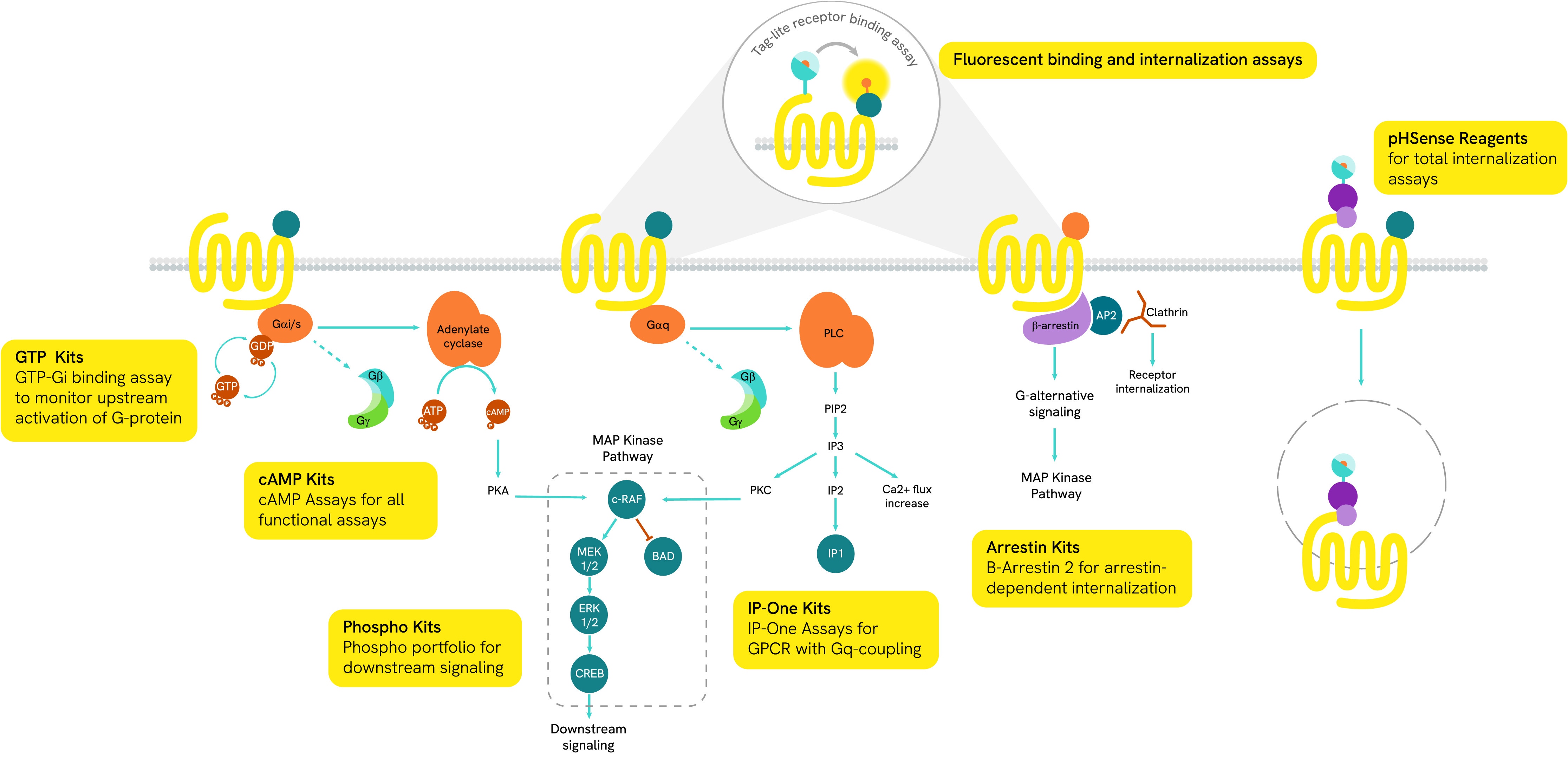
The ligand/receptor binding is the first key step in GPCR signaling. Revvity offers a range of ligand/receptor interaction solutions in both radioactive format and fluorescence assays with our proprietary HTRF-based Tag-lite technology.
The ligand/receptor binding is the first key step in GPCR signaling. Revvity offers a range of ligand/receptor interaction solutions in both radioactive format and fluorescence assays with our proprietary HTRF-based Tag-lite technology.
-
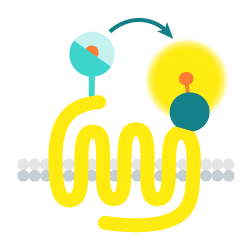
Tag-lite - Tag-lite is a non-radioactive TR-FRET-based solution which can be used to assess the pharmacology and pharmacodynamics of ligand/receptor interactions. The receptor of interest is labeled with cryptate in a way that does not alter receptor binding. The corresponding ligand is labeled with d2 acceptor. When the d2-labeled ligand binds to the cryptate-labeled receptor, it produces a HTRF signal.
In a competition assay, the introduction of a competing small molecule dislodges the d2-labeled ligand. Consequently, the ligand is pushed away and the signal stops.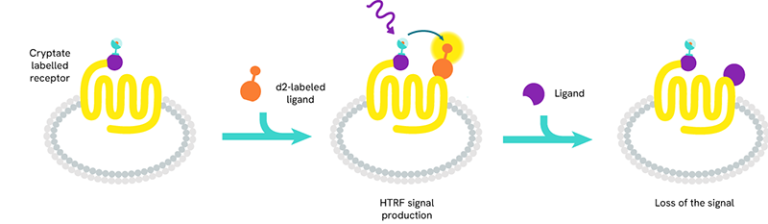
-
Receptor ligand binding with radiolabeled ligands and GPCR membranes - We also offer a range of radioactive ligands, which are used to study molecular interactions and QC our GPCR over-expressing cell lines and membrane preparations. Radiometric ligand binding assays are conducted on cells or cell membranes containing a GPCR receptor of interest. Radioligands can be used to perform saturation curves, competition, and kinetic experiments.
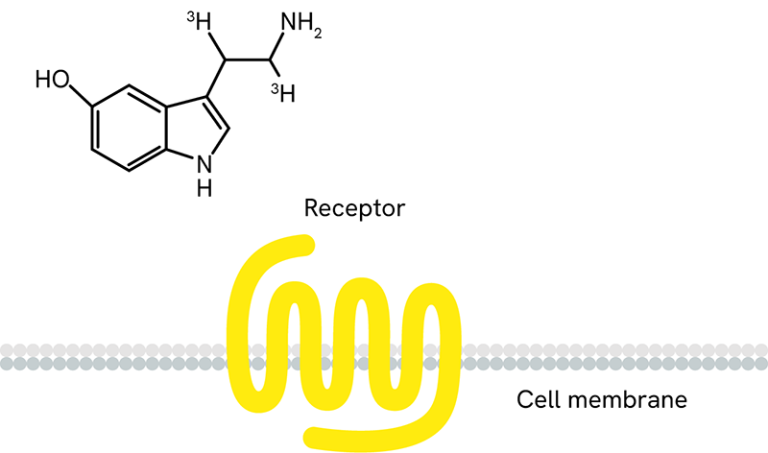
- Radioactive ligand binding assays can be performed in several different formats. Typically, we perform this assay in filtration format, where the unbound ligand is washed away using a vacuum manifold or cell harvester. The assay can also be conducted in a homogenous Scintillation Proximity Assay (SPA) format, where no wash steps are required.
Over 100 GPCR-expressing membrane preparations are validated for receptor ligand binding and over 50 are validated for GTPγS binding.
G-protein activation
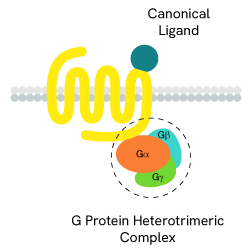
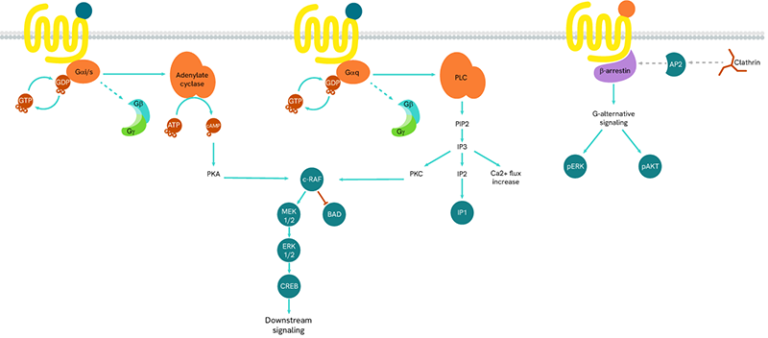
The main signal transduction of GPCRs is dependent on the receptor-mediated activation of heterotrimeric G-proteins.
The main signal transduction of GPCRs is dependent on the receptor-mediated activation of heterotrimeric G-proteins.
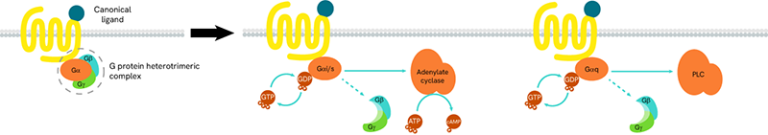
G-proteins are composed of three subunits (Gα, Gβ and Gγ) and are classified into four families (Gs, Gi/o, Gq/11, and G12/13):
- Gαs-coupled GPCRs positively stimulate the activity of adenylate cyclase, resulting in an increase in cellular cAMP
- Gαi-coupled GPCRs lead to a negative regulation of adenylate cyclase, and thus to a decrease in cAMP production
-
Gαq protein signaling is based on enzymes of the phospholipase C family (PLC).

Arrestins recruitment
GPCRs also initiate G-protein-independent pathways that instead rely on arrestin coupling, which in turn suppresses G-protein activation.
GPCRs also initiate G-protein-independent pathways that instead rely on arrestin coupling, which in turn suppresses G-protein activation.
When a ligand binds to the extracellular part of a GPCR, the receptor undergoes conformational changes and opens its intracellular part to be phosphorylated by kinases. This phosphorylation of GPCRs by GRKs (GPCR kinases) is a prerequisite for high-affinity arrestin binding. The phosphorylated GPCR becomes a binding site for arrestins. Once linked, arrestins can interact with several partners such as AP2 or signaling proteins.

Intracellular signaling
GPCRs are at the beginning of many phosphorylation cascades involving phosphatases bound to second messengers.
GPCRs are at the beginning of many phosphorylation cascades involving phosphatases bound to second messengers.
Following the activation of a GPCR, signals are sent via G-proteins and arrestins to second messengers. Each GPCR has its own signaling pathway, but kinases and phosphatases are always involved and promote phosphorylation cascades to the nucleus. These molecular events are implicated in many physiological and cellular processes such as cell survival, proliferation, differentiation, and metabolism.

GPCR Internalization Assays
G protein-coupled receptor (GPCR) internalization plays a critical role in regulating receptor availability at the cell surface, contributing to desensitization and controlling the duration and intensity of GPCR-mediated signaling.
G protein-coupled receptor (GPCR) internalization plays a critical role in regulating receptor availability at the cell surface, contributing to desensitization and controlling the duration and intensity of GPCR-mediated signaling.
Revvity's pHSense assay offers a robust, plate-reader-based solution for monitoring GPCR internalization. Utilizing a pH-sensitive dye, the assay is optimized for kinetic and time-resolved measurements, even at low endogenous expression levels. It enables rapid, high-throughput data generation using standard plate readers, making it ideal for drug discovery workflows.
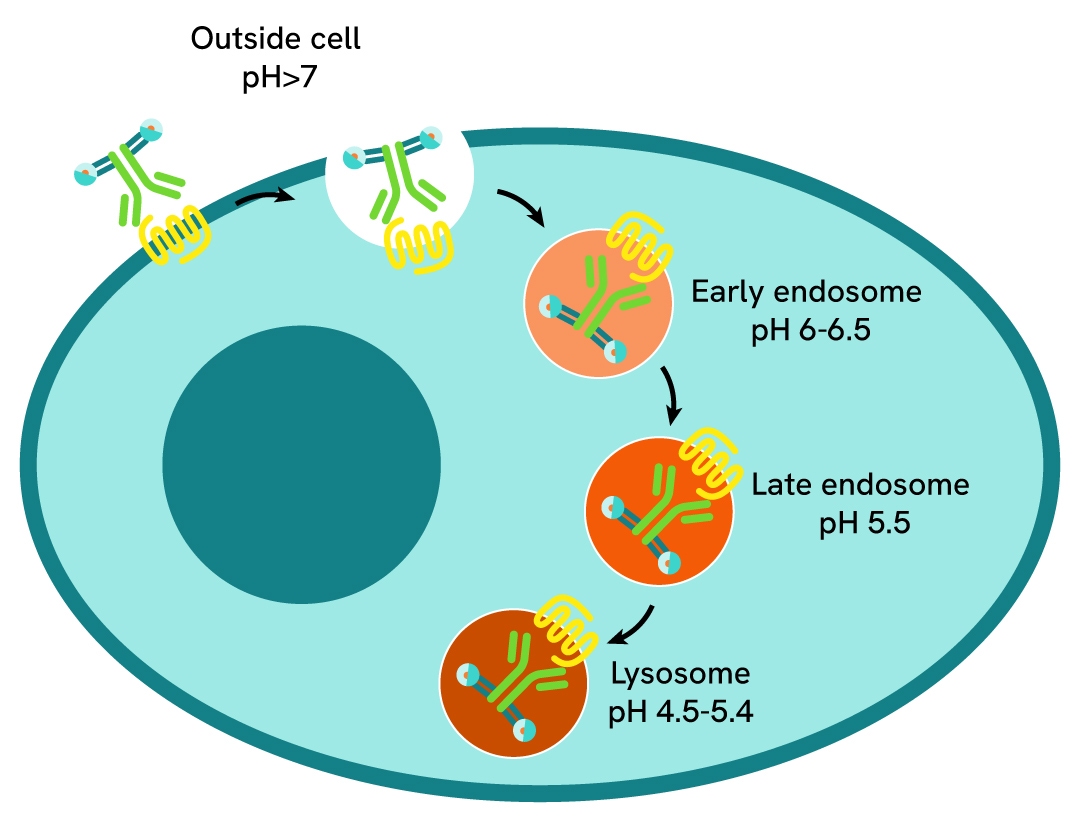
In pHSense assay, a pH-sensitive probe is directly (SNAP-labeling) or indirectly (Probe-labeled anti-tag or Probe-labeled Fab + anti-GPCR) bound to the receptor of interest. At neutral pH (extracellular pH), the probe is minimally fluorescent. As receptors are internalized, they enter acidic cellular compartments (endosomes, late endosomes, lysosomes) where the probe starts emitting a strong and long-lived fluorescent signal.
pHSense SNAP labeling Reagent

pHSense Anti-Flag Antibody

Internalization of SNAP-Flag-GPCR in HEK293 cells
HEK293 cells that overexpress SNAP-Flag-GLP1R were plated at 80K cells/well and incubated 1H at 23°C with either pHSense SNAP labeling Reagent or pHSense Anti-Flag Antibody. Cells were then incubated 20mn at 37°C with increasing concentrations of GLP1R agonist Exendin 4.
The results show dose-dependent internalization responses of GLP1R in presence of Exendin 4,in line with the published literature.


Internalization of endogenous GPCRs in unmodified cells
Rat insulinoma beta cells (INS-1E) were plated at 25K cells/well and cultured 96 hours. The internalization assay was run in culture medium. Cells were pre-incubated for 1H at 23°C with a 2:1 mix of pHSense Fab Anti-Mouse IgG and anti-GLP1R antibody. Following this pre-incubation, cells were either incubated 1H at 37°C with agonist Exendin 4, or incubated 30mn with antagonist Exendin 9-39 then 1H with Exendin 4.
The results show a dose-dependent internalization of GLP1R in presence of Exendin 4 alone, while the pre-incubation with Exendin 9-39 neutralizes this agonist-induced internalization and results in internalization levels identical to that of the receptor constitutive activity.GLP1R internalization.
pHSense Fab Anti-Mouse IgG + Anti-GPCR


Read more about GPCR internalization assays in our dedicated technical note Technical Note phsense eu probes time-resolved fluorescence plate based applications for monitoring internalization of overexpressed and endogenous GPCRs .
Featured resources