

HTRF Human Phospho-Phospholamban (Ser16) Detection Kit, 10,000 Assay Points






This HTRF kit enables the cell-based quantitative detection of phosphorylated Phospholamban at Ser16.
| Feature | Specification |
|---|---|
| Application | Cell Signaling |
| Sample Volume | 16 µL |
This HTRF kit enables the cell-based quantitative detection of phosphorylated Phospholamban at Ser16.









Product information
Overview
This cell-based assay enables the detection of Phosphorylated Phospholamban (PLN) at Serine 16. The phospho assay along with the total PLN assay are ideal for monitoring the activation of PKA in response GPCR stimulation, such as the beta2 Adrenergic receptor. PKA phosphorylates PLN at the Ser 16 residue.
In an unphosphorylated state, PLN inhibits SERCA2a which is the Ca2+ pump controling the contractility of the cardiac muscle. PLN phosphorylation induced by PKA and CaMKII, on Ser16 and Thr17 sites respectively, abrogates SERCA2a inhibition. The PLN phosphorylation state regulates the activity of this Ca2+ pump in the heart.
How it works
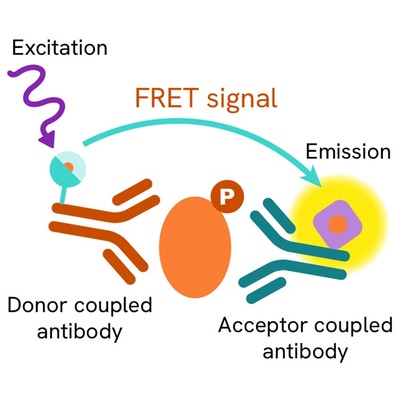
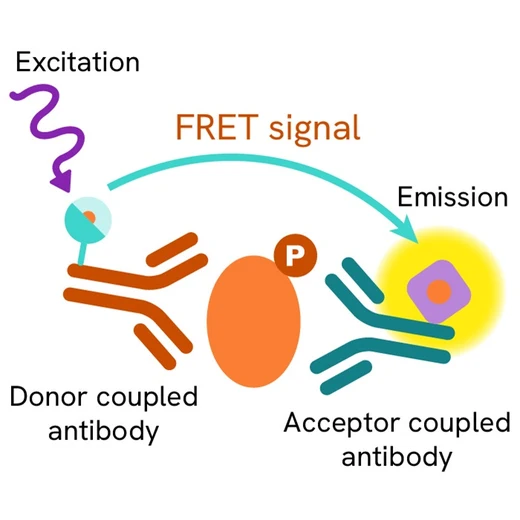
Phospho-Phospholamban (Ser16) assay principle
The Phospho-Phospholamban (Ser16) assay measures Phospholamban when phosphorylated at Ser16. Unlike Western Blot, the assay is entirely plate-based and does not require gels, electrophoresis, or transfer. The Phospho-Phospholamban (Ser16) assay uses 2 labeled antibodies: one with a donor fluorophore, the other one with an acceptor. The first antibody is selected for its specific binding to the phosphorylated motif on the protein, the second for its ability to recognize the protein independent from its phosphorylation state. Protein phosphorylation enables an immune-complex formation involving both labeled antibodies, and which brings the donor fluorophore into close proximity to the acceptor, thereby generating a FRET signal. Its intensity is directly proportional to the concentration of phosphorylated protein present in the sample, and provides a means of assessing the protein’s phosphorylation state under a no-wash assay format.

Phospho-Phospholamban (Ser16) 2-plate assay protocol
The 2 plate protocol involves culturing cells in a 96-well plate before lysis, then transferring lysates to a 384-well low volume detection plate before adding Phospho-Phospholamban (Ser16) HTRF detection reagents. This protocol enables the cells' viability and confluence to be monitored.

Phospho-Phospholamban (Thr17)1-plate assay protocol
Detection of Phosphorylated Phospholamban (Ser16)with HTRF reagents can be performed in a single plate used for culturing, stimulation, and lysis. No washing steps are required. This HTS designed protocol enables miniaturization while maintaining robust HTRF quality.

Assay validation
Phospho Ser16-phospholamban assay on heart homogenate
Spontaneously hypertensive (SHR) heart and wistar Kyoto (WKY) heart samples were homogenized using Cisbio lysis buffer, and supernatant was collected after centrifugation. Total protein were quantified and a side by side dilution was done in lysis buffer. 16 µL of lysate were transferred into a 384-well sv white microplate, and 4 µL of the HTRF phospho-Phospholamban (Ser16) detection reagents were added. The HTRF signal was recorded after an overnight incubation at room temperature.

Phospho-phospholamaban comparison on hypertensive rat heart lysates
Spontaneously hypertensive (SHR) heart and wistar Kyoto (WKY) heart samples were homogenized using Cisbio lysis buffer, and supernatant was collected after centrifugation. On a fixed total protein concentration (16.25 µg/mL), a side by side comparison was done between the HTRF phospho Trh17 PLN kit and the phospho-Ser16 PLN kit. In order to normalize the detection, the total phospholamban was also detected. 16 µL of lysate were transferred into a 384-well sv white microplate, and 4 µL of the HTRF phospho-Phospholamban (Ser16) detection reagents or the phospho-Phospholamban (Thr17) detection reagents or total phospholamban were added. The HTRF signal was recorded after an overnight incubation at room temperature. The data is presented after a normalization on the total expression level.
**** t-test analysis P value <0.0001

HTRF phospho Ser16-PLN assay compared to Western Blot
SHR heart homogenates were collected after centrifuging for 10 minutes. Equal amounts of total were used for a side by side comparison of WB and HTRF. 110 µg/mL of total protein were used as the highest concentration of a serial dilution carried out. HTRF phospho-PLN assay shows better sensitivity compared to WB, as 0.49µg/mL of total protein was sufficient to detect the phosphorylation, much lower than 13.3 µg/mL for WB.

Simplified pathway
Simplified pathway for PLN
Phospholamban (PLN) plays a crucial role in heart failure through its control of cardiac sarcoplasmic reticulum (SR) Ca2+-ATPase (SERCA2a). This protein has a Ca2+ pump included in the SR membrane, and once activated, Ca2+ goes inside SR. Insufficient SERCA2a activity is a hallmark of heart failure. Dephosphorylated PLN inhibits SERCA2a, whereas phosphorylation of PLN at either the Ser16 site by PKA or the Thr17 site by CaMKII reverses the inhibition of SERCA2a. The PLN phosphorylation state regulates the activity of this Ca2+ pump. Relaxation is driven by dephosphorylation of PLN, and contraction by the phosphorylation status.
This small protein, is present in cardiac, smooth, and slow-twitch skeletal muscles. However, its regulatory effects have mainly been studied in cardiac muscle. The activation process is not well known, but 2 main pathways are described ending in 2 different phosphaorylations: on residue Serine 16, or on Threonine 17. Binding of a β-agonist to its receptor activates G protein, which enhances adenylate cyclase (AC) activity. AC catalyzes cAMP formation, which activates PKA. PKA phosphorylates the L-type Ca2+ channel, increasing the Ca2+ influx, and phosphorylates PP1 and the Ser16 residue of PLN. An increase in intracellular Ca2+ causes the activation of CaMKII. This autophosphorylation state is also controlled by PP1. CaMKII in turn phosphorylates PLN at the Thr17 residue.

Specifications
| Application |
Cell Signaling
|
|---|---|
| Brand |
HTRF
|
| Detection Modality |
HTRF
|
| Lysis Buffer Compatibility |
Lysis Buffer 1
Lysis Buffer 2
Lysis Buffer 3
Lysis Buffer 4
Lysis Buffer 5
|
| Molecular Modification |
Phosphorylation
|
| Product Group |
Kit
|
| Sample Volume |
16 µL
|
| Shipping Conditions |
Shipped in Dry Ice
|
| Target |
Phospholamban
|
| Target Class |
Phosphoproteins
|
| Target Species |
Human
|
| Technology |
TR-FRET
|
| Therapeutic Area |
Cardiovascular
|
| Unit Size |
10,000 assay points
|
Video gallery
Resources
Are you looking for resources, click on the resource type to explore further.
This guide provides you an overview of HTRF applications in several therapeutic areas.


Loading...
How can we help you?
We are here to answer your questions.






























