

HTRF Human and Mouse Phospho-IRS1 (mouse Ser307/human Ser312) Detection Kit, 500 Assay Points






| Feature | Specification |
|---|---|
| Application | Cell Signaling |
| Sample Volume | 16 µL |









Product information
Overview
This HTRF cell-based assay conveniently and accurately quantifies phosphorylated IRS1 at (Ser307 mouse/Ser312 human). IRS1 is a relevant marker for insulin resistance, tumor development, and metastatic progression. It is associated with type 2 diabetes, obesity, and cancer.
HTRF assays offer many advantages over other technologies:
- Homogeneous add-and-read format
- No wash steps
- Low background
- Straightforward miniaturization from 96- or 384-well microplates to high density assay formats such as 384-well low volume and 1536-well plates
- Stable signal, providing flexibility in time of readout or size of assays
How it works
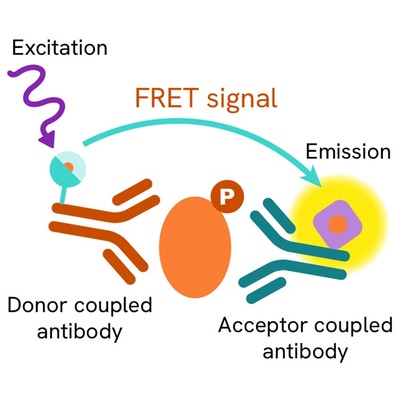
Phospho-IRS1 assay principle
The phospho-IRS1(Ser307 mouse/Ser312 human) assay measures IRS when phosphorylated at Ser307 mouse/Ser312 human. Contrary to Western Blot, the assay is entirely plate-based and does not require gels, electrophoresis or transfer. The phospho-IRS1(Ser307 mouse/Ser312 human) assay uses 2 labeled antibodies: one with a donor fluorophore, the other one with an acceptor. The first antibody is selected for its specific binding to the phosphorylated motif on the protein, the second for its ability to recognize the protein independent of its phosphorylation state. Protein phosphorylation enables an immune-complex formation involving both labeled antibodies and which brings the donor fluorophore into close proximity to the acceptor, thereby generating a FRET signal. Its intensity is directly proportional to the concentration of phosphorylated protein present in the sample, and provides a means of assessing the protein’s phosphorylation state under a no-wash assay format.

Phospho-IRS1 2-plate assay protocol
The 2 plate protocol involves culturing cells in a 96-well plate before lysis then transferring lysates to a 384-well low volume detection plate before adding Phospho-IRS1 HTRF detection reagents. This protocol enables the cells' viability and confluence to be monitored.

Phospho-IRS1 1-plate assay protocol
Detection of phosphorylated IRS1 with HTRF reagents can be performed in a single plate used for culturing, stimulation and lysis. No washing steps are required. This HTS designed protocol enables miniaturization while maintaining robust HTRF quality.

Assay validation
Phospho-IRS1 assay validation on C2C12 mouse myotubes
C2C12 cells were cultured until 100% confluency was reached. They were then cultured for 7 days in 2% horse serum media to induce the differentiation. After a starvation step in serum-free medium, cells were treated with a cocktail of pro-inflammatory cytokines and/or Insulin. After cell supernatant removal, cells were lysed with 1.5 mL of supplemented lysis buffer #1 for 30 min at RT under gentle agitation. 16 µL of lysate were then transferred into a 384-well low volume white microplate for HTRF detection using the phospho- and total IRS1 kit reagents.


Phospho-IRS1 assay validation on 3T3-L1 mouse adipocytes
3T3-L1 cells were cultured in medium with 10% newborn calf serum. They were then cultured for 7 days in medium containing 10% FCS, insulin, dexamethasone, IBMX and µM thiazolidinedione to induce differentiation from fibroblasts to adipocytes. After an overnight starvation step in serum-free medium, the cells were treated with 100 nM insulin for 45 min or with a cocktail of pro-inflammatory cytokines for 30 min. After cell supernatant removal, cells were lysed and transferred into a 384-well low volume white microplate for HTRF detection using the phospho- and total IRS1 kit reagents (Samples were kindly provided by JF Tanti’s research team, UMR INSERM U1065/UNS, C3M, Nice, France).

Phospho-IRS1 assay validation on MCF-7 human breast cancer cells
50,000 human breast cancer MCF-7 cells were plated in a 96-well plate in complete culture medium, and incubated for 24h at 37°C, 5% CO2. Increasing concentrations of Insulin were added and incubated for 45 min at 37°C. After cell culture medium removal, cells were lysed with 50 µL of lysis buffer for 30 min at RT under gentle shaking. 16 µL of lysate were then transferred into a 384-well sv white microplate for detection of phospho-IRS1 Ser 312 and total IRS1, using 4 µL of the HTRF detection reagents.

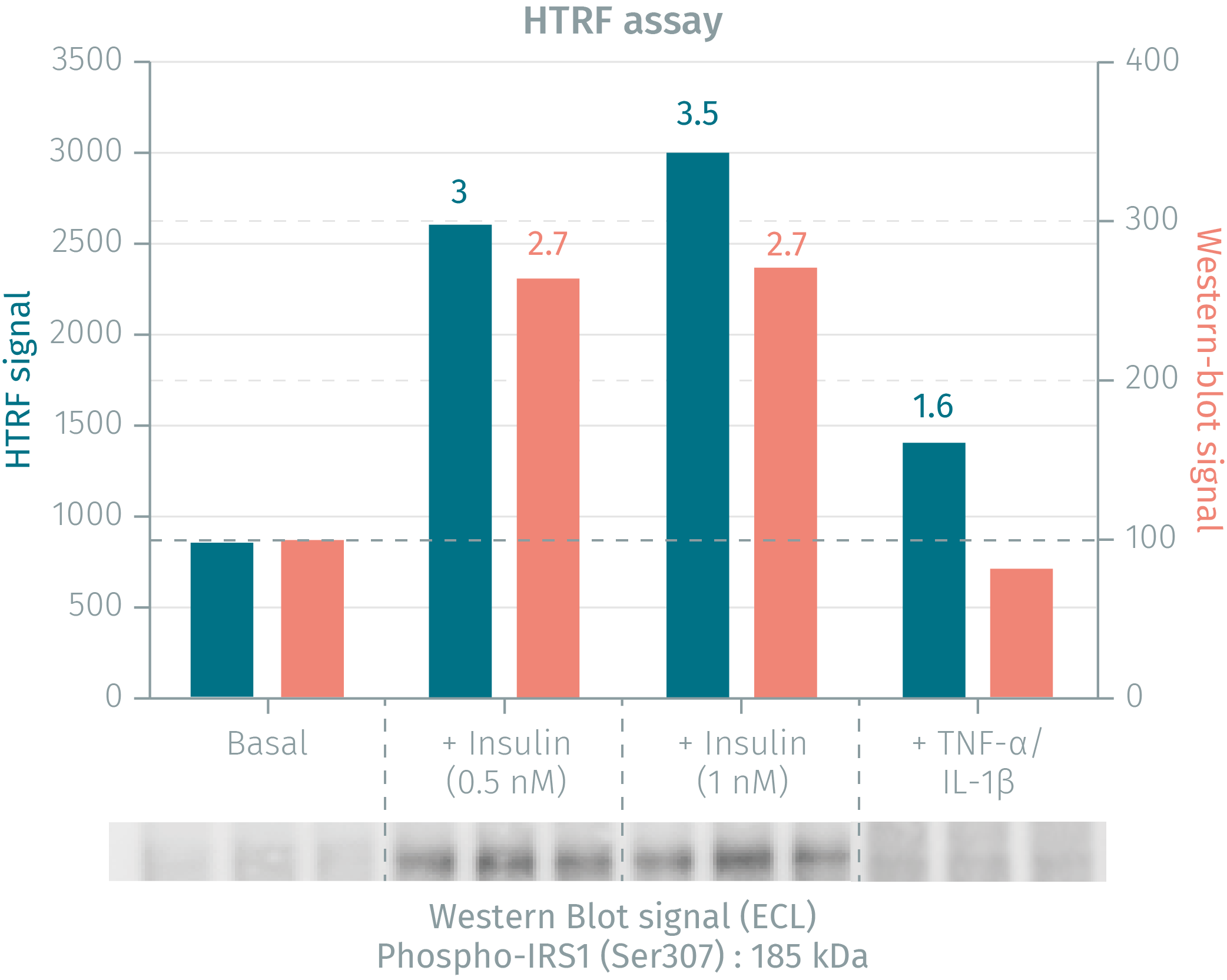
HTRF phospho-IRS1 assay compared to Western Blot
3T3-L1 mouse fibroblasts were plated in 12-well plates and cultured at 37°C, 7% CO2 in complete culture medium with 10% newborn calf serum. Cells were then cultured for 7 days in medium containing 10% FCS, 5 µg/mL insulin, 0.25 µM dexamethasone, 0.5 mM IBMX and 10 µM thiazolidinedione to induce the differentiation from fibroblasts to adipocytes. After an overnight starvation step in serum-free medium, the cells were treated with insulin for 45 min or with a cocktail of pro-inflammatory cytokines (10 ng/mL TNF-α + 10 ng/mL IL-1β) for 30 min. After cell supernatant removal, cells were lysed with 600 µL of supplemented lysis buffer #1 for 30 min at RT under gentle agitation. After a 10 min centrifugation step, 16 µL of lysate were transferred in triplicate into a 384-well plate for HTRF detection or into a gel for WB detection. Data presented on the graph correspond to the mean of each triplicate. The HTRF assay provides higher pharmacological assay windows than Western Blot.
Simplified pathway
IRS1/PI3K/AKT signaling pathway

Modulation of the IRS1/PI3K/AKT signaling pathway
Inhibition of the pathway. While tyrosine-phosphorylated IRS-1 positively regulates insulin signaling, serine phosphorylation generally serves as negative feedback to down-regulate IRS-1 function. Increased serine phosphorylation of IRS-1 represents a hallmark of insulin resistance in adipose tissue and skeletal muscles. Binding of Insulin or IGF-1 to the extracellular a-subunits of the IR or IGF-1R triggers autophosphorylation of the transmembrane ß-subunits of the receptor, thus activating it. IRS1/AKT pathway activation plays a critical role in maintaining glucose homeostasis. AKT also activates mTOR that mediates cell growth and survival, as well as protein synthesis. IRS1 downregulation is a relevant diabetic marker while overexpression is associated with metastatic growth and cancers.
Activation of the pathway. Binding of Insulin or IGF-1 to the extracellular a-subunits of the IR or IGF-1R (or to hybrids of both receptors) triggers autophosphorylation of the transmembrane ß-subunits of the receptor. IRS-1 is then recruited by the activated tyrosine kinase receptor and phosphorylated on multiple tyrosine residues, creating a docking site for PI3K at the membrane. The PI3K pathway is then activated with downstream phosphorylations of AKT, followed by AS160, GSK3 and FoxO1, leading respectively to GLUT4 translocation/glucose uptake, glycogen synthesis and inhibition of gluconeogenesis. Thus, the IRS1/AKT pathway activation plays a critical role in maintaining glucose homeostasis. AKT also activates mTOR that mediates cell growth and survival, as well as protein synthesis.
Related disorders
IRS1 phosphorylation and degradation, which lead to the inactivation of the Insulin/IRS1 pathway, make it a relevant marker for insulin resistance in skeletal muscles cells and adipocytes, while upregulation of the IGF-1/IRS1 pathway upon IRS1 overexpression, is a readout for tumor development and metastatic progression.
Specifications
| Application |
Cell Signaling
|
|---|---|
| Brand |
HTRF
|
| Detection Modality |
HTRF
|
| Lysis Buffer Compatibility |
Lysis Buffer 1
|
| Molecular Modification |
Phosphorylation
|
| Product Group |
Kit
|
| Sample Volume |
16 µL
|
| Shipping Conditions |
Shipped in Dry Ice
|
| Target Class |
Phosphoproteins
|
| Target Species |
Human
Mouse
|
| Technology |
TR-FRET
|
| Therapeutic Area |
Metabolism/Diabetes
NASH/Fibrosis
|
| Unit Size |
500 assay points
|
Video gallery
Resources
Are you looking for resources, click on the resource type to explore further.
Discover the versatility and precision of Homogeneous Time-Resolved Fluorescence (HTRF) technology. Our HTRF portfolio offers a...
This guide provides you an overview of HTRF applications in several therapeutic areas.
This flyer details HTRF™ and AlphaLISA™ assays for investigating key biomarkers and signaling pathways in metabolic disease...
Obesity is a complex condition characterized by excessive fat accumulation, posing significant health and socioeconomic challenges...


Loading...
How can we help you?
We are here to answer your questions.






























