
DELFIA Eu-TDA Cytotoxicity Kit
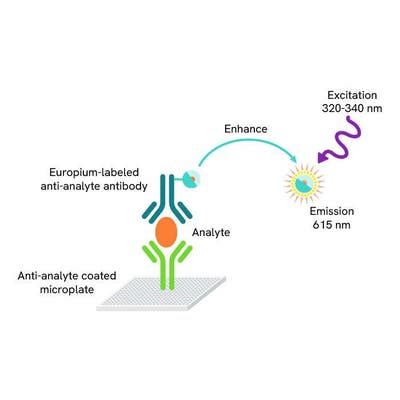
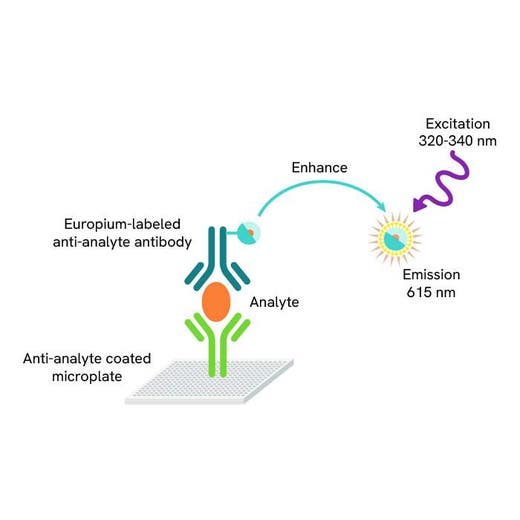
DELFIA® TRF (time-resolved fluorescence) cytotoxicity kit for measuring chemical cytotoxicity, cell-mediated cytotoxicity, and antibody-dependent cell cytotoxicity (ADCC). This kit has the same assay principle as a chromium release assay, with target cells loaded with a BATDA ligand instead of 51Cr. There is no requirement for specially-engineered cells.
| Feature | Specification |
|---|---|
| Application | Protein Quantification |
| Sample Volume | 200 µL |
DELFIA® TRF (time-resolved fluorescence) cytotoxicity kit for measuring chemical cytotoxicity, cell-mediated cytotoxicity, and antibody-dependent cell cytotoxicity (ADCC). This kit has the same assay principle as a chromium release assay, with target cells loaded with a BATDA ligand instead of 51Cr. There is no requirement for specially-engineered cells.


Loading...
Product information
Overview
DELFIA time-resolved fluorescence cell cytotoxicity assays offer a non-radioactive method to Chromium 51, for use in cell-mediated cytotoxicity and antibody-dependent cell cytotoxicity (ADCC) studies. The method is based on loading cells with an acetoxymethyl ester of a fluorescence enhancing ligand. After the ligand has penetrated the cell membrane the ester bonds are hydrolyzed within the cell to form a hydrophilic ligand, which no longer passes through the membrane. After cytolysis the released ligand is introduced to a europium solution to form a fluorescent chelate. The measured signal correlates directly with the amount of lysed cells.
Reagents for the assay are also available separately:
- Lysis buffer (30 mL), #4005-0010
- DELFIA Eu-Solution (200 mL), #C135-100
- DELFIA BATDA labeling reagent (50 µL), #C136-100
Selected references:
- Lazar, G. A. et al. Engineered antibody Fc variants with enhanced effector function. PNAS 103, 4005-4010 (2006).
- Tsai, C.-Y. et al. Type I IFNs and IL-18 Regulate the Antiviral Response of Primary Human γδ T Cells against Dendritic Cells Infected with Dengue Virus. The Journal of Immunology 194, 3890-3900 (2015).
- Mehta, R. S., Chen, X., Antony, J., Boyiadzis, M. & Szabolcs, P. Generating Peripheral Blood Derived Lymphocytes Reacting Against Autologous Primary AML Blasts. J Immunother 39, 71-80 (2016).
- Snyder, K. M. et al. Expression of a Recombinant High Affinity IgG Fc Receptor by Engineered NK Cells as a Docking Platform for Therapeutic mAbs to Target Cancer Cells. Front Immunol 9, (2018).
- Yu, T., Qiao, C., Lv, M. & Tang, L. Novel anti-CD38 humanized mAb SG003 possessed enhanced cytotoxicity in lymphoma than Daratumumab via antibody-dependent cell-mediated cytotoxicity. BMC Biotechnology 19, 28 (2019).
Specifications
| Application |
Protein Quantification
|
|---|---|
| Automation Compatible |
Yes
|
| Brand |
DELFIA
|
| Detection Modality |
DELFIA
|
| Product Group |
Kit
|
| Sample Volume |
200 µL
|
| Shipping Conditions |
Shipped in Blue Ice
|
| Target Class |
Biomarkers
|
| Technology |
TRF
|
| Unit Size |
960 assay points
|
Resources
Are you looking for resources, click on the resource type to explore further.
Discover how Revvity's no-wash immunoassay technologies - including HTRF and AlphaLISA - streamline CMC processes for biologic...
DELFIA® immunoassays are particularly well suited for discovery of specific, high-affinity monoclonal antibodies (mAbs)...
DELFIA immunoassays are a superior alternative to traditional ELISAs
Discover expert guidelines for optimizing cell labeling in DELFIA TRF cytotoxicity assays. Learn how to enhance assay performance...
Loading...


How can we help you?
We are here to answer your questions.






























