

HTRF Human and Mouse Phospho-VASP (Ser239) Detection Kit, 500 Assay Points




The phospho-VASP (Ser239) enables the cell-based quantitative detection of VASP phosphorylated on Ser 239 for monitoring PKA and PKG activation.
| Feature | Specification |
|---|---|
| Application | Cell Signaling |
| Sample Volume | 16 µL |
The phospho-VASP (Ser239) enables the cell-based quantitative detection of VASP phosphorylated on Ser 239 for monitoring PKA and PKG activation.






Loading...
Product information
Overview
Phospho-VASP (Ser239) cellular assay is ideal for monitoring PKG activation, which phosphorylates VASP on Serine 239. VASP is involved in cell motility, migration, and adhesion. This makes phospho-VASP assaying a valuable tool in cardiovascular, oncology, and inflammation research.
HTRF assays offer many advantages over other technologies:
- Homogeneous add-and-read format
- No wash steps
- Low background
- Straightforward miniaturization from 96- or 384-well microplates to high density assay formats such as 384-well low volume and 1536-well plates
- Stable signal, providing flexibility in time of readout or size of assays
How it works
Phospho-VASP (Ser239) assay principle
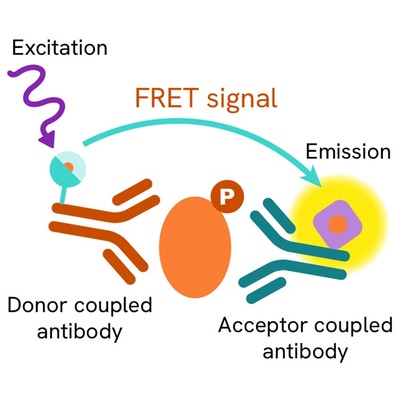
The Phospho-VASP (Ser239) assay measures VASP when phosphorylated at Ser239. Contrary to Western Blot, the assay is entirely plate-based and does not require gels, electrophoresis or transfer. The Phospho-VASP (Ser239) assay uses 2 labeled antibodies: one with a donor fluorophore, the other one with an acceptor. The first antibody is selected for its specific binding to the phosphorylated motif on the protein, the second for its ability to recognize the protein independent of its phosphorylation state. Protein phosphorylation enables an immune-complex formation involving both labeled antibodies and which brings the donor fluorophore into close proximity to the acceptor, thereby generating a FRET signal. Its intensity is directly proportional to the concentration of phosphorylated protein present in the sample, and provides a means of assessing the proteins phosphorylation state under a no-wash assay format.

Phospho-VASP (Ser239) 2-plate assay protocol
The 2 plate protocol involves culturing cells in a 96-well plate before lysis then transferring lysates to a 384-well low volume detection plate before adding phospho-VASP (Ser239) HTRF detection reagents. This protocol enables the cells' viability and confluence to be monitored.

Phospho-VASP (Ser239) 1-plate assay protocol
Detection of Phosphorylated VASP (Ser239) with HTRF reagents can be performed in a single plate used for culturing, stimulation and lysis. No washing steps are required. This HTS designed protocol enables miniaturization while maintaining robust HTRF quality.

Assay validation
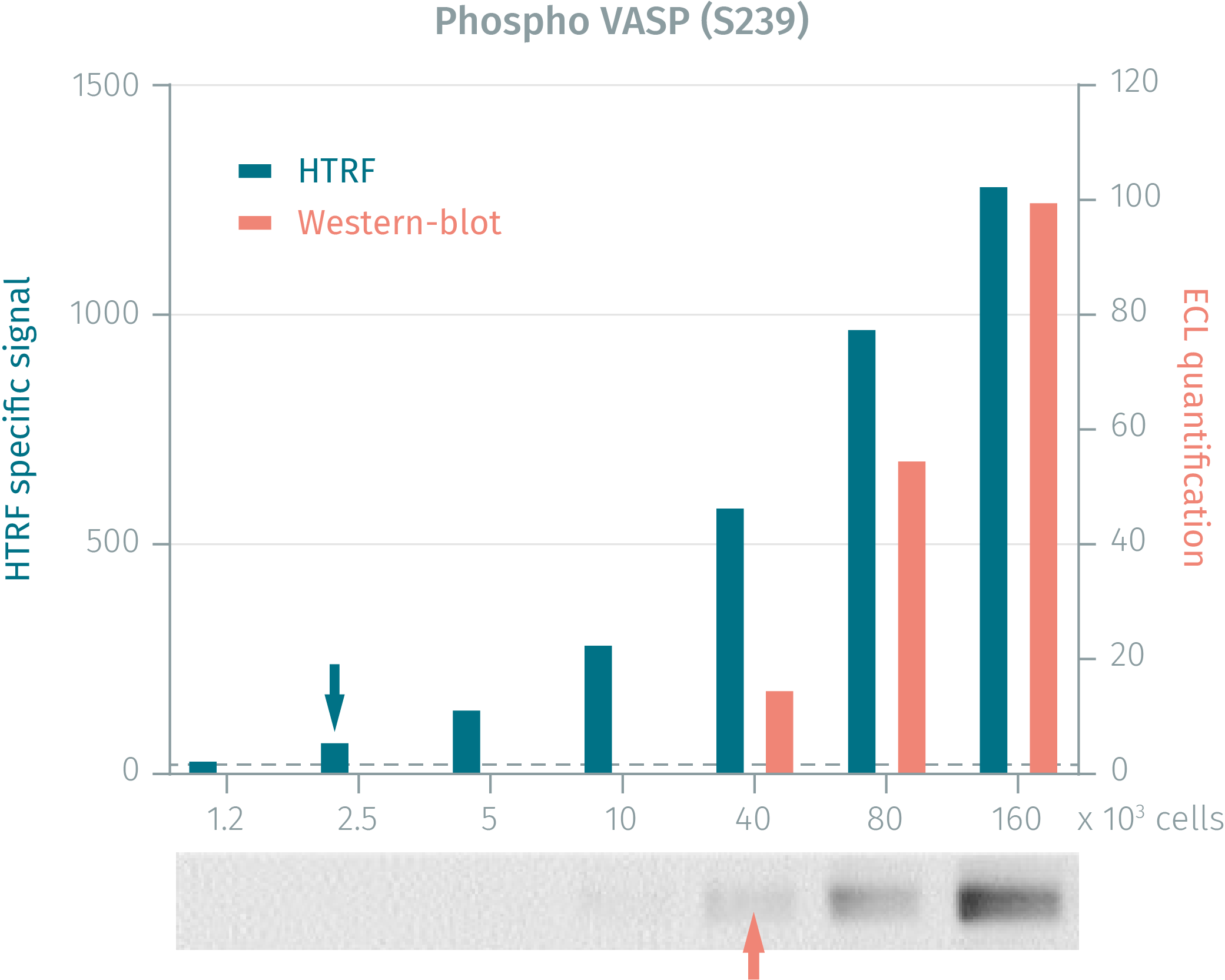
HTRF assay compared to western blot using phospho-VASP (s239) assay on mouse NIH3T3 cells
Mouse NIH3T3 cells were plated at 8.106 cells in a T175 cm2 flask and incubated for 48h at 37°C, 5% CO2. After 50µM forskolin treatment for 30 minutes, the cells were lysed with 3mL of lysis buffer for 30min at RT under gentle shaking. Serial dilutions of the cell lysate were performed in the supplemented lysis buffer, and 16 µL of each dilution were transferred into a 384-well low volume white microplate before the addition of 4 µL of the HTRF phospho-VASP (Ser239) detection reagents. Equal amounts of lysates were used for a side by side comparison of Western Blot and HTRF. For HTRF assays, 2 500 cells were enough for minimal signal detection, while 40 000 cells were needed for a Western Blot signal detection. Thus, phospho-VASP (Ser239) HTRF assays is at least 4-fold more sensitive than the Western Blot.

Validation on A431cells treated with forskolin
Human A431 cells were plated at 100,000 cells/well in a 96 well plate, and incubated for 24h at 37°C, 5% CO2. After treatment for 30 min with increasing concentrations of Forskolin, the medium was removed and the cells were lysed with 60 µL of lysis buffer for 30min at RT under gentle shaking. 16 µL of lysate were transferred into a 384-well low volume white microplate and 4 µL of the HTRF phospho-VASP (Ser157), phosphor-VASP (S239) or total VASP detection reagents were added. The HTRF signal was recorded after an overnight incubation.

Validation on NIH3T3 cells treated with forskolin
Mouse NIH3T3 cells were plated at 100,000 cells/well in a 96 well plate, and incubated for 24h at 37°C, 5% CO2. After treatment for 30 min with increasing concentrations of Forskolin, the medium was removed and the cells were lysed with 60 µL of lysis buffer for 30 min at RT under gentle shaking. 16 µL of lysate were transferred into a 384-well low volume white microplate and 4 µL of the HTRF phospho-VASP (Ser157), phosphor-VASP (S239) or total VASP detection reagents were added. The HTRF signal was recorded after an overnight incubation.
Simplified pathway
Function and regulation of VASP
Vasodilator-stimulated phosphoprotein (VASP) is an actin-associated protein and a member of the Ena-VASP family. VASP stimulates actin filament elongation, and is involved in cytoskeleton remodeling and cell polarity. VASP proteins are involved in axon guidance, platelet activation and cell migration. In platelets, VASP is a major substrate for cAMP-dependent protein kinase A (PKA) and cGMP-dependent protein kinase G (PKG). Whereas the preferred site for PKA is Ser-157, the preferred site for PKG is Ser-239. Phosphorylation modulates F-actin binding, actin filament elongation and platelet activation. VASP phosphorylation is often used to monitor the effect of drugs that reduce platelet reactivity in cardiovascular diseases.

Specifications
| Application |
Cell Signaling
|
|---|---|
| Brand |
HTRF
|
| Detection Modality |
HTRF
|
| Lysis Buffer Compatibility |
Lysis Buffer 1
Lysis Buffer 2
Lysis Buffer 4
Lysis Buffer 5
|
| Molecular Modification |
Phosphorylation
|
| Product Group |
Kit
|
| Sample Volume |
16 µL
|
| Shipping Conditions |
Shipped in Dry Ice
|
| Target Class |
Phosphoproteins
|
| Target Species |
Human
Mouse
|
| Technology |
TR-FRET
|
| Therapeutic Area |
Cardiovascular
Metabolism/Diabetes
|
| Unit Size |
500 assay points
|
Video gallery
Citations
Resources
Are you looking for resources, click on the resource type to explore further.
Atherosclerosis pathogenesis, cellular actors, and pathways
Atherosclerosis is a common condition in which arteries harden and...
Discover the versatility and precision of Homogeneous Time-Resolved Fluorescence (HTRF) technology. Our HTRF portfolio offers a...
This guide provides you an overview of HTRF applications in several therapeutic areas.
Loading...


How can we help you?
We are here to answer your questions.






























