
HTRF Human and Mouse Phospho-STAT3 (Tyr705) High Performance Detection Kit, 500 Assay Points

This High Performance HTRF kit enables cell-based quantitative detection of STAT3 phosphorylated at Tyr705, delivering higher signal-to-background ratios and improved sensitivity for more robust and reliable results.
| Feature | Specification |
|---|---|
| Application | Cell Signaling |
| Sample Volume | 16 µL |
This High Performance HTRF kit enables cell-based quantitative detection of STAT3 phosphorylated at Tyr705, delivering higher signal-to-background ratios and improved sensitivity for more robust and reliable results.


Loading...
Product information
Overview
This High Performance HTRF cell-based assay enables rapid, quantitative, and highly sensitive detection of STAT3 phosphorylated on Tyr705 as a readout of JAK/STAT signaling activity. Compared to the standard version, the High-Performance format delivers a higher signal-to-background ratio and improved sensitivity, ensuring more robust results even in challenging conditions. These enhancements make it possible to obtain reliable results on more physiological models such as primary cells, broadening the applicability of the assay for translational and advanced research. STAT3 is upregulated in various diseases, particularly autoimmune disorders, neurodegenerative diseases, or cancers, making this kit a powerful tool for therapeutic strategies by inhibiting the pathway.
HTRF assays offer many advantages over other technologies:
- Homogeneous add-and-read format
- No wash steps
- Low background
- Straightforward miniaturization from 96- or 384-well microplates to high density assay formats such as 384-well low volume and 1536-well plates
- Stable signal, providing flexibility in readout time or size of assays
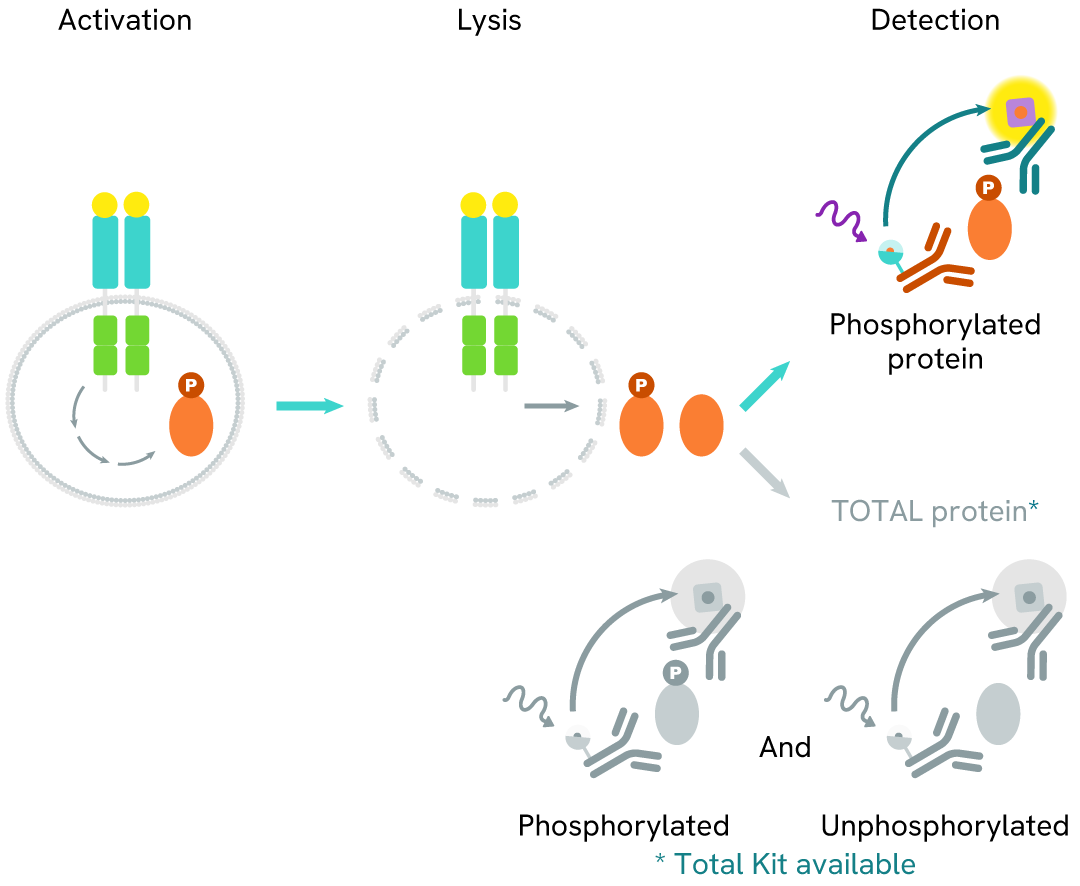
How it works
High Performance Phospho-STAT3 (Tyr705) assay principle
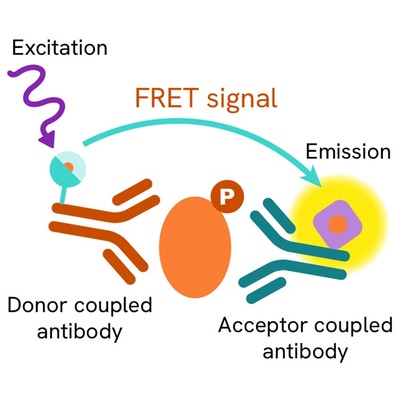
The High Performance phospho-STAT3 (Tyr705) assay measures STAT3 when phosphorylated at Tyr705. Unlike Western Blot, the assay is entirely plate-based and does not require gels, electrophoresis, or transfer. It uses two antibodies: one labeled with a donor fluorophore and the other with an acceptor. The first antibody specifically binds the phosphorylated motif on the protein, while the second recognizes the protein independently of its phosphorylation state. Protein phosphorylation enables the formation of an immune complex involving both labeled antibodies, bringing the donor fluorophore into close proximity to the acceptor and generating a FRET signal. Its intensity is directly proportional to the concentration of phosphorylated protein in the sample, allowing assessment of the protein’s phosphorylation state in a no-wash assay format.

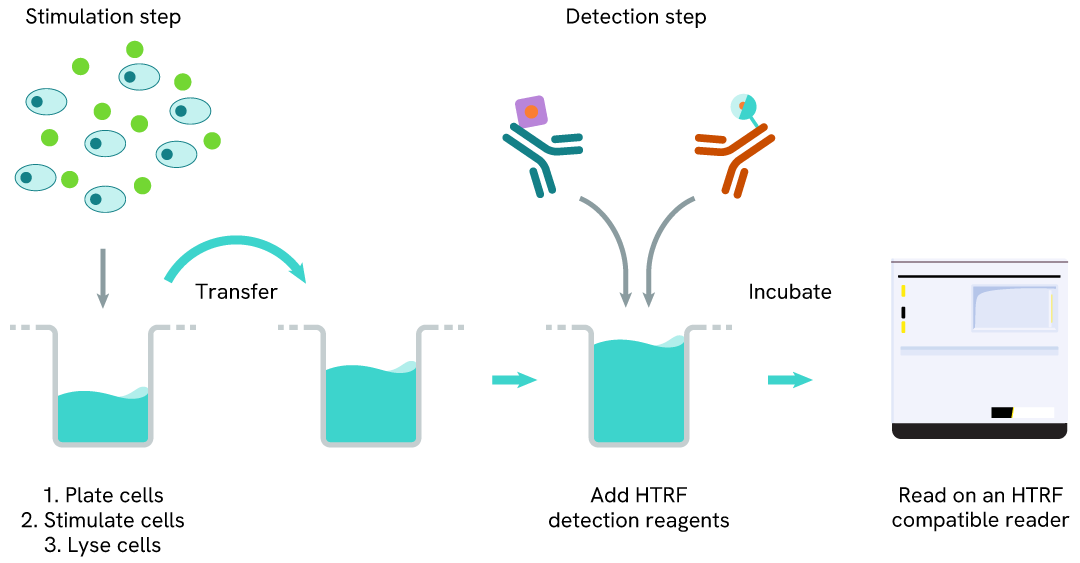
High Performance Phospho-STAT3 (Tyr705) two-plate assay protocol
The two-plate protocol involves culturing cells in a 96-well plate before lysis, then transferring lysates into a 384-well low volume detection plate before the addition of High Performance phospho-STAT3 (Tyr705) HTRF detection reagents. This protocol allows the cells’ viability and confluence to be monitored.
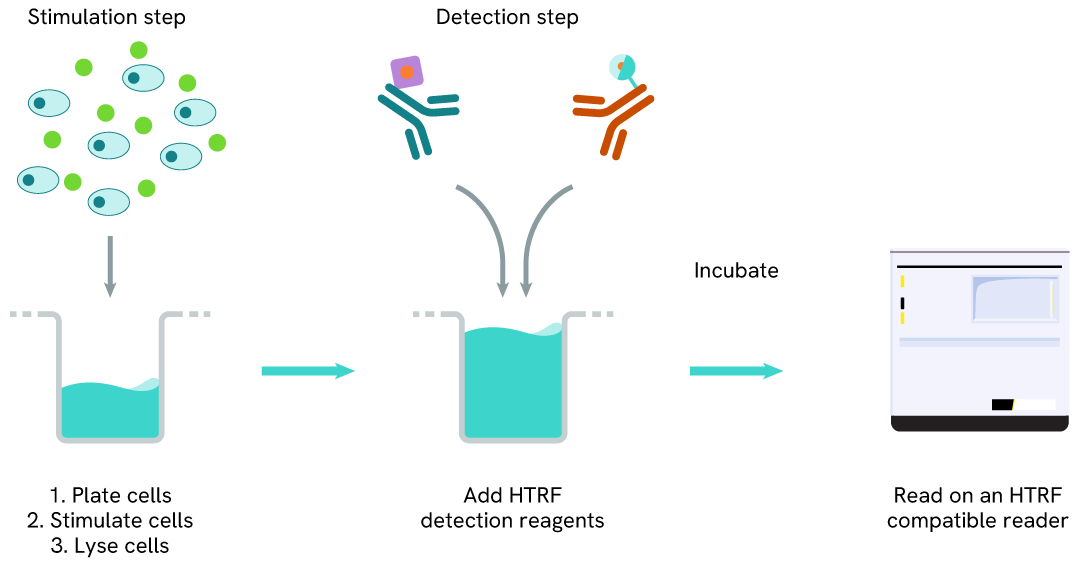
High Performance Phospho-STAT3 (Tyr705) one-plate assay protocol
Detection of phosphorylated STAT3 (Tyr705) with HTRF reagents can be performed in a single plate used for culturing, stimulation, and lysis. No washing steps are required. This HTS designed protocol allows miniaturization while maintaining robust HTRF quality.
Assay validation
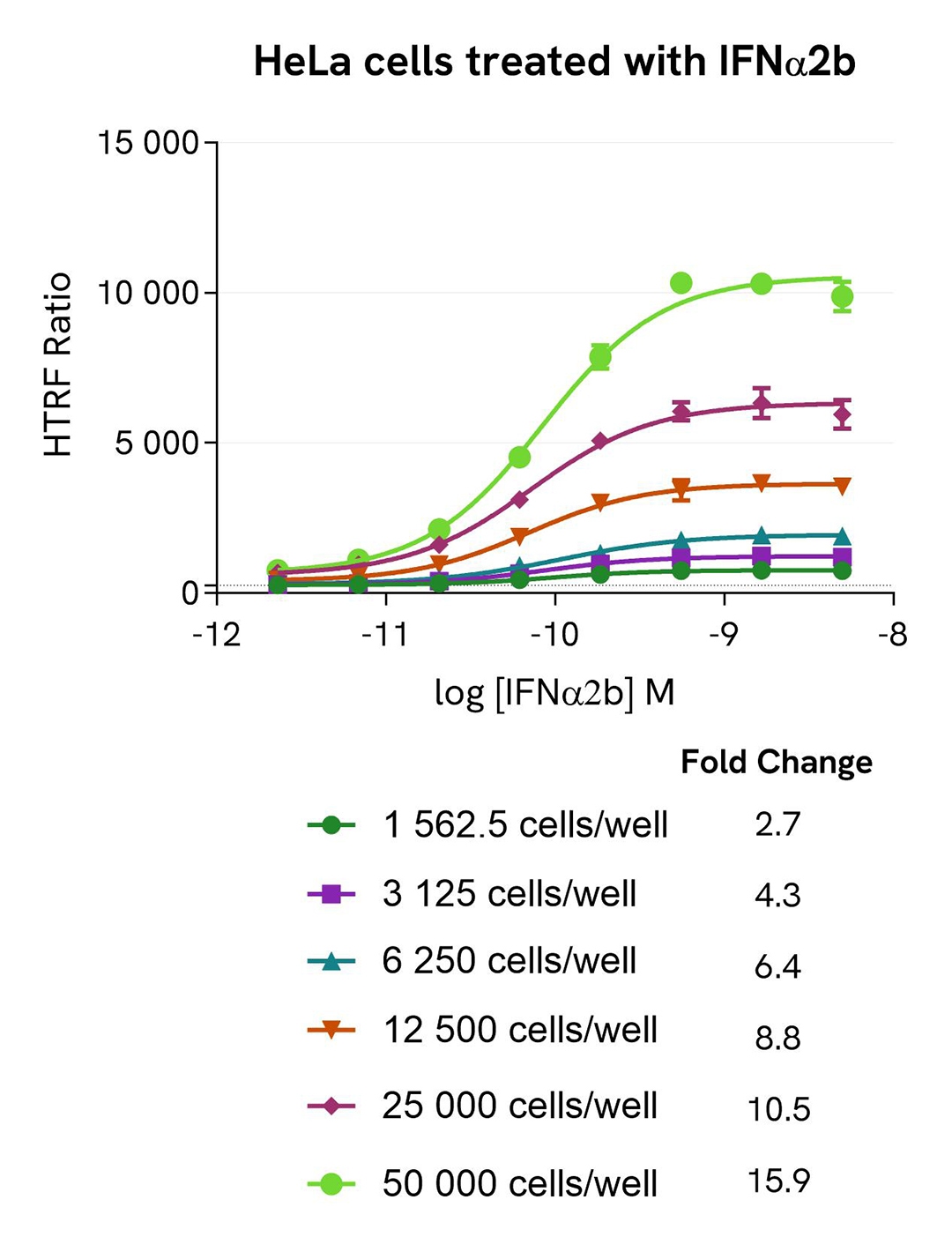
High-sensitivity detection of STAT3 phosphorylation induced by IFNα2b dose response in HeLa cells
HeLa cells were seeded at various densities in a 96-well culture-treated plate with complete culture medium, and incubated overnight at 37 °C, 5% CO₂. The cells were then treated for 30 minutes with increasing concentrations of IFNα2b.
After treatment, the cells were lysed with 50 µL of supplemented lysis buffer #1 for 30 minutes at room temperature under gentle shaking. For the detection step, 16 µL of cell lysate were transferred into a 384-well low-volume white microplate, and 4 µL of the HTRF High Performance Phospho-STAT3 (Tyr705) detection reagents were added. The HTRF signal was recorded after 2 hours of incubation.
As expected, IFNα2b triggered a dose-dependent increase in Phospho-STAT3 (Tyr705) levels, detectable with high sensitivity, and significantly measurable at around 1,500 cells per well.
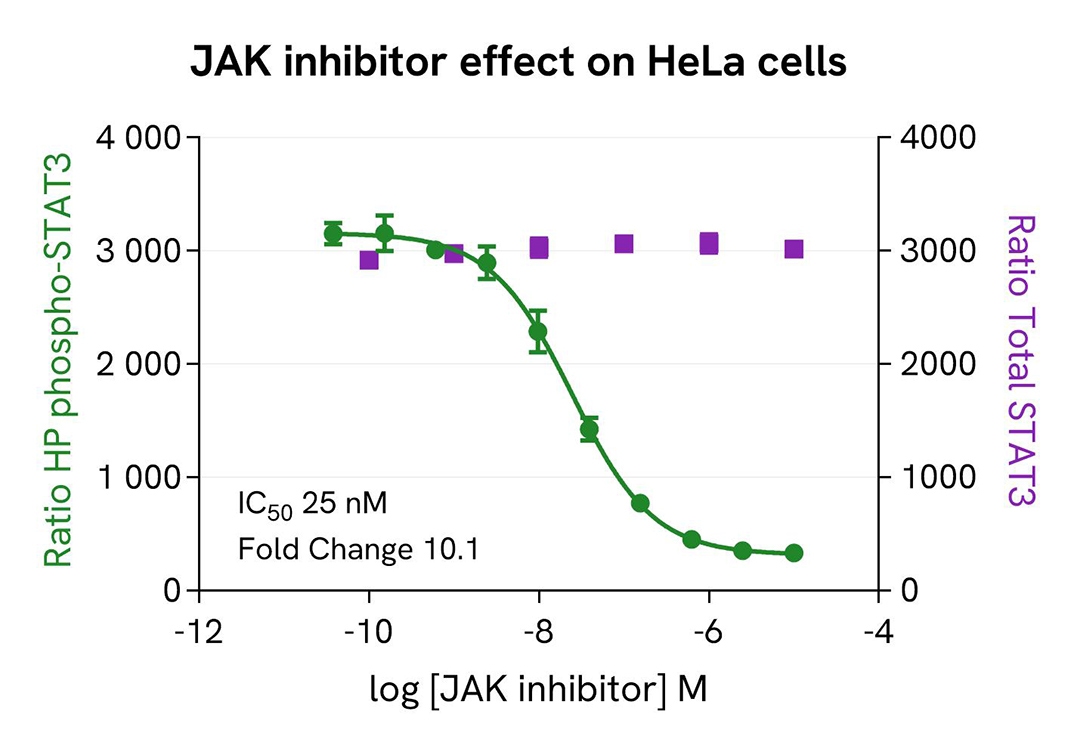
Inhibition of Phospho-STAT3 (Tyr705) by a JAK inhibitor in HeLa cells
HeLa cells were seeded in a 96-well culture-treated plate (25,000 cells/well) in complete culture medium, and incubated overnight at 37 °C, 5% CO₂. The following day, the cells were treated for 30 minutes with increasing concentrations of a JAK inhibitor. Then the agonist IFNα2b (0.2 nM) was added for an additional hour before the end of the treatment.
The cells were lysed with 50 µL of supplemented lysis buffer #1 for 30 minutes at room temperature under gentle shaking. For the detection step, 16 µL of cell lysate were transferred into a 384-well low-volume white microplate, and 4 µL of the HTRF High Performance Phospho-STAT3 (Tyr705) or Total-STAT3 detection reagents were added. The HTRF signal was recorded after 2 hours for the Phospho assay and after overnight incubation for the Total assay.
As expected, the JAK inhibitor induced a dose-dependent decrease in STAT3 phosphorylation, whereas the expression level of Total STAT3 remained unchanged, and no toxicity was detected (ATPlite Luminescence Assay System, #6016943).
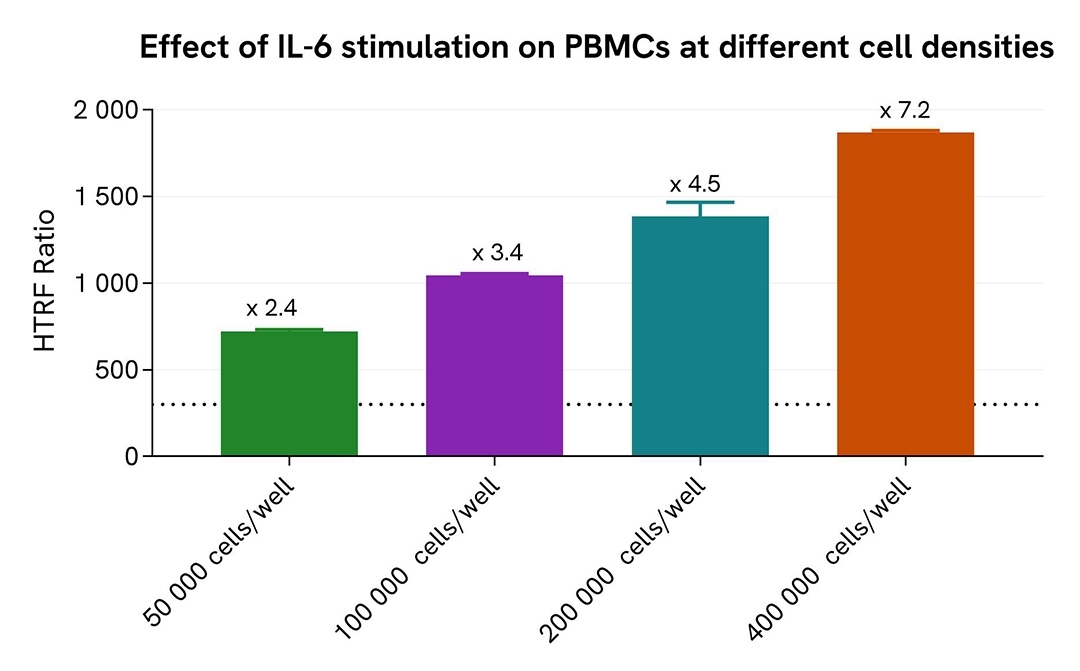
Pharmacological Validation in a PBMC model
Human PBMCs were seeded at 50,000 to 400,000 cells per well in a 96-well culture-treated plate, and stimulated with hIL-6 (1 µg/mL) for 15 minutes. After stimulation, the cells were lysed with 10 µL of supplemented lysis buffer #1 (4X) for 30 minutes at room temperature under gentle shaking.
For the detection step, 16 µL of cell lysate were transferred into a 384-well low-volume white microplate, and 4 µL of the HTRF High Performance Phospho-STAT3 (Tyr705) detection reagents were added. The HTRF signal was recorded after 2 hours of incubation.
As expected, IL-6 stimulation triggered an increase in Phospho-STAT3 (Tyr705) levels, detectable with high sensitivity from as few as 50,000 cells per well in this model.
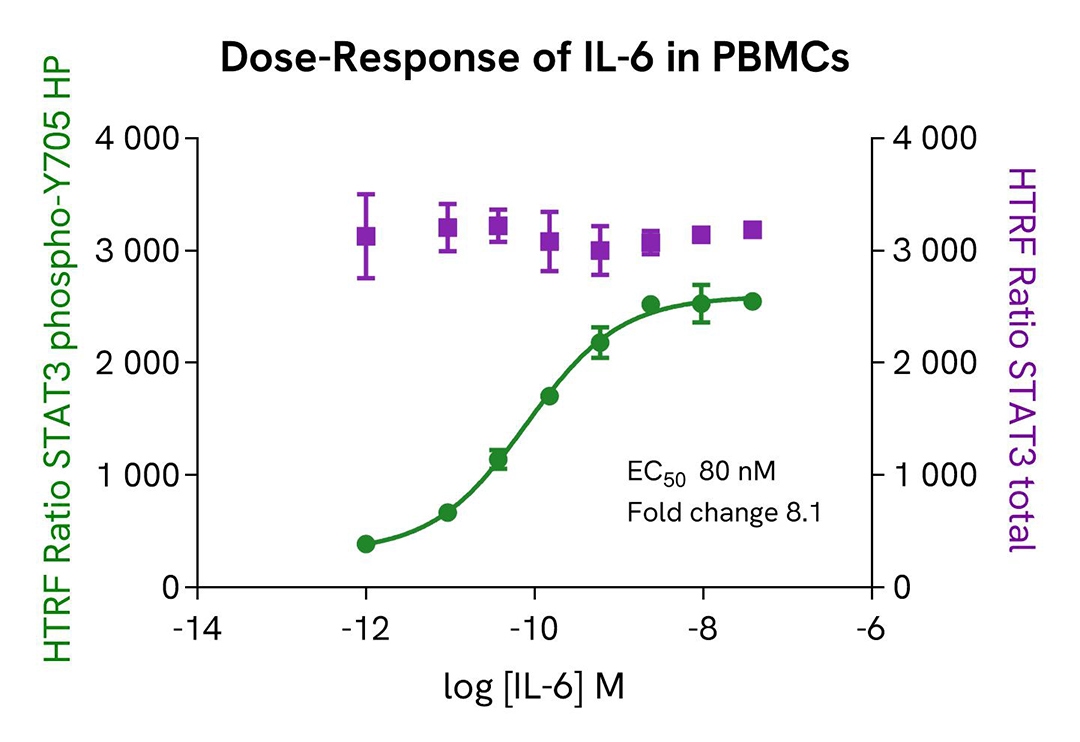
Pharmacological validation in a PBMC model
Human PBMCs were plated at 400,000 cells per well in a 96-well culture-treated plate with 25 µL of medium, and stimulated with increasing concentrations of hIL-6 (5 µL) for 15 minutes. After stimulation, the cells were lysed with 10 µL of supplemented lysis buffer #1 (4X) for 30 minutes at room temperature under gentle shaking.
For the detection step, 16 µL of cell lysate were transferred into a 384-well low-volume white microplate, and 4 µL of the HTRF High Performance Phospho-STAT3 (Tyr705) or Total-STAT3 detection reagents were added. The HTRF signal was recorded after 2 hours for the Phospho assay and after overnight incubation for the Total assay.
As expected, IL-6 stimulation triggered a dose-dependent increase in Phospho-STAT3 (Tyr705) levels, detectable with high sensitivity in a model lacking basal STAT3 phosphorylation.
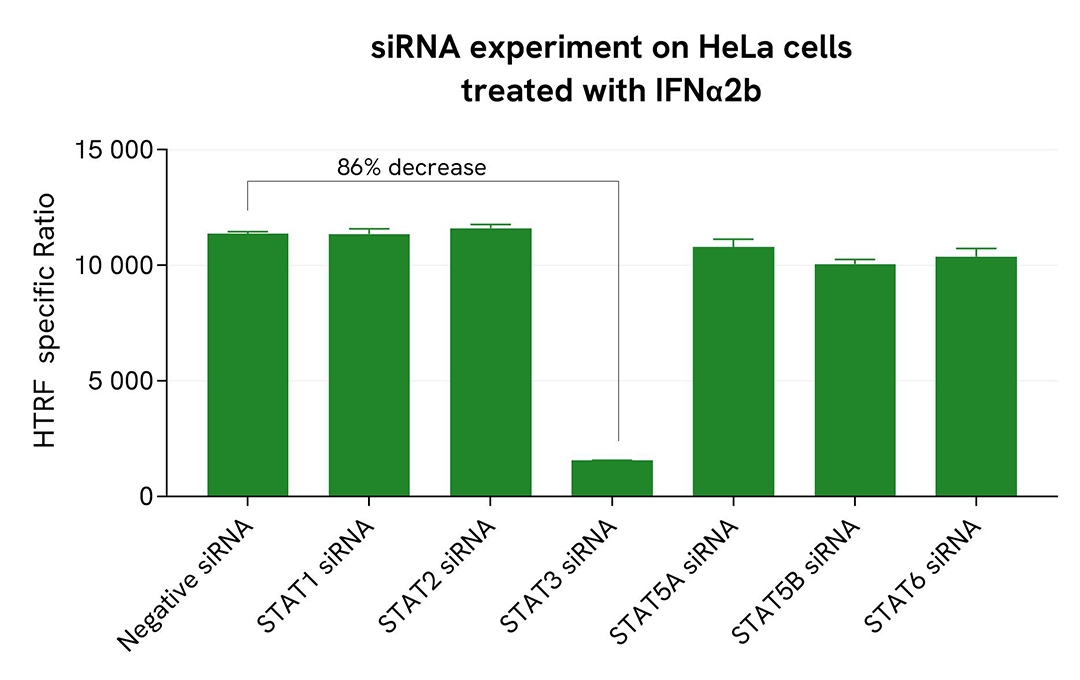
Validation of High Performance Phospho-STAT3 (Tyr705) assay specificity by siRNA knockdown experiments
HeLa cells were plated in a 96-well plate (25,000 cells per well) and cultured for 24 hours. The cells were then transfected with siRNAs specific for STAT1, STAT2, STAT3, STAT5A, STAT5B, or STAT6, as well as a negative control, siRNA. After 48 hours of incubation, the cells were treated with IFNα2b (5 nM) for 30 minutes before lysis.
For detection, 16 µL of lysate were transferred into a 384-well low-volume white microplate, and 4 µL of the HTRF High Performance Phospho-STAT3 (Tyr705) detection reagents were added. The HTRF signal was recorded after 3 hours of incubation.
Transfection with STAT3 siRNA led to an 86% decrease in signal compared to cells transfected with the negative control siRNA. In contrast, knockdown of STAT1, STAT2, STAT5A, STAT5B, and STAT6 did not induce any signal decrease, demonstrating that the HTRF High Performance Phospho-STAT3 (Tyr705) assay is specific for STAT3 phosphorylation and does not cross-react with other STAT family members.
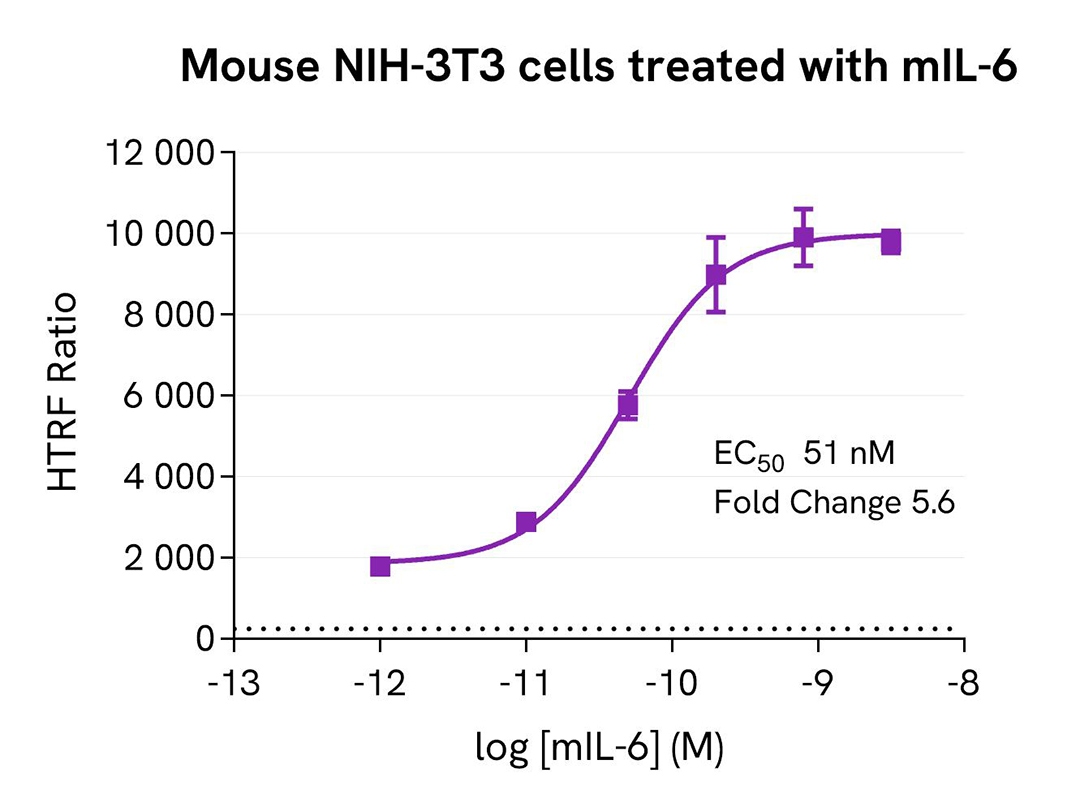
Cross-reactivity assessment in mouse NIH-3T3 cells
Mouse NIH-3T3 cells were seeded at 100,000 cells per well in a 96-well microplate. After 24 hours of incubation, the cells were treated for 30 minutes with increasing concentrations of mIL-6, then lysed for 30 minutes using supplemented lysis buffer #1 at room temperature under gentle shaking.
16 µL of lysate were transferred into a 384-well low-volume white microplate, followed by the addition of 4 µL of HTRF Phospho-STAT3 (Tyr705) detection reagents. The HTRF signal was recorded after a 2-hour incubation.
The HTRF Phospho-STAT3 (Tyr705) assay detected phosphorylation stimulation in mouse NIH-3T3 cells, which displayed a high basal level of phospho-STAT3 (Tyr705), demonstrating good compatibility with mouse samples.
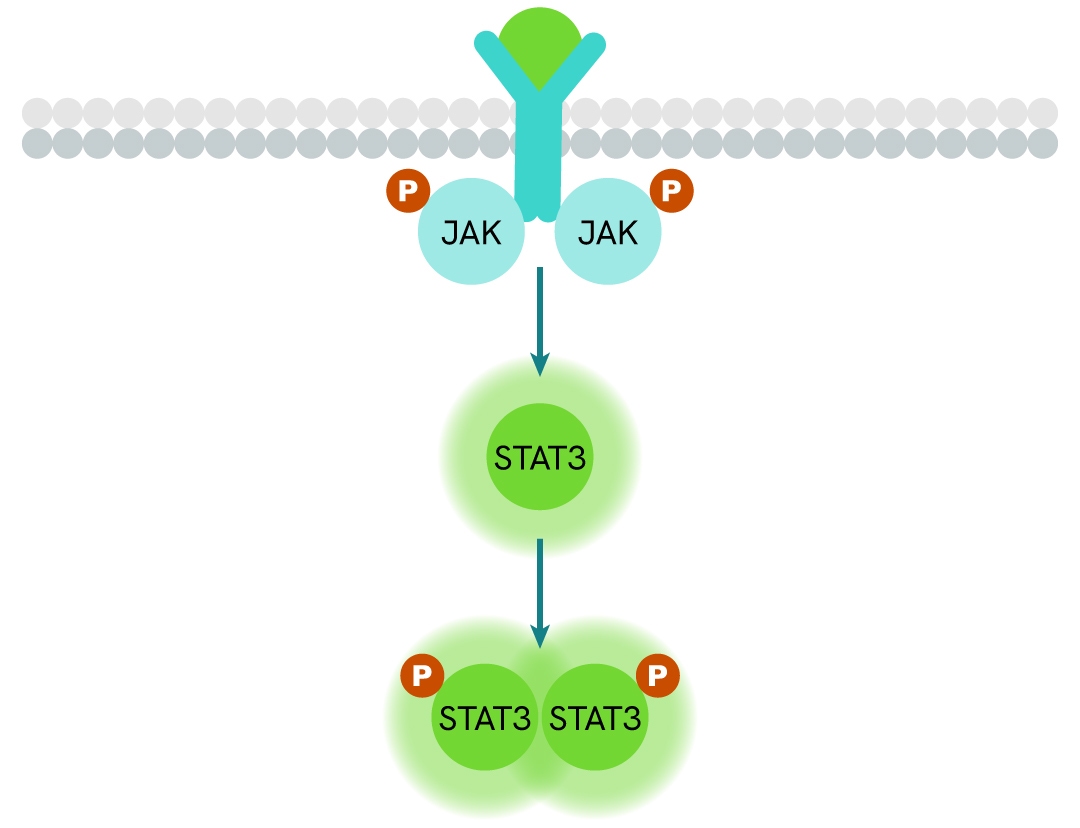
Simplified pathway
STAT3 signaling pathway
In response to cytokines and growth factors, STAT3 is phosphorylated by receptor-associated kinases and subsequently forms dimers that translocate to the cell nucleus, where they function as transcriptional activators. STAT3 regulates the expression of numerous genes in response to cellular stimuli and plays a critical role in processes such as cell growth and apoptosis. Binding of Interleukin-6 family cytokines to the gp130 receptor triggers STAT3 phosphorylation via JAK2. STAT3 is also activated by other receptors, including receptor tyrosine kinases (RTKs) such as EGFR and c-Met, as well as by c-Src.
Specifications
| Application |
Cell Signaling
|
|---|---|
| Brand |
HTRF
|
| Detection Modality |
HTRF
|
| Lysis Buffer Compatibility |
Lysis Buffer 1
Lysis Buffer 3
Lysis Buffer 4
|
| Molecular Modification |
Phosphorylation
|
| Product Group |
Kit
|
| Sample Volume |
16 µL
|
| Shipping Conditions |
Shipped in Dry Ice
|
| Target |
STAT3
|
| Target Class |
Phosphoproteins
|
| Target Species |
Human
Mouse
|
| Technology |
TR-FRET
|
| Unit Size |
500 assay points
|
Resources
Are you looking for resources, click on the resource type to explore further.
Loading...


How can we help you?
We are here to answer your questions.






























