
HTRF Human and Mouse TREM2 (Ectodomain) Detection Kit, 10,000 Assay Points
The HTRF Ectodomain TREM2 kit is designed for the rapid detection and quantification of the TREM2 receptor by targeting its ectodomain in cellular lysates.
| Feature | Specification |
|---|---|
| Application | Protein Quantification |
| Dynamic Range | 81 - 12,000 pg/mL |
| Limit of Detection | 9 pg/mL |
| Limit of Quantification | 21 pg/mL |
| Sample Volume | 16 µL |
The HTRF Ectodomain TREM2 kit is designed for the rapid detection and quantification of the TREM2 receptor by targeting its ectodomain in cellular lysates.


Loading...
Product information
Overview
The kit is designed for the rapid detection of the TREM2 receptor by targeting its ectodomain in cellular lysates. TREM2 (Triggering Receptor Expressed on Myeloid Cells 2) belongs to the TREM family of receptors, which regulate the activity of various immune cell types, including neutrophils, monocytes/macrophages, microglia, and dendritic cells.
HTRF assays offer many advantages over other technologies:
- Homogeneous add-and-read format
- No wash steps
- Low background
- Straightforward miniaturization from 96- or 384-well microplates to high density assay formats such as 384-well low volume and 1536-well plates
- Stable signal, providing flexibility in readout time or size of assays
How it works
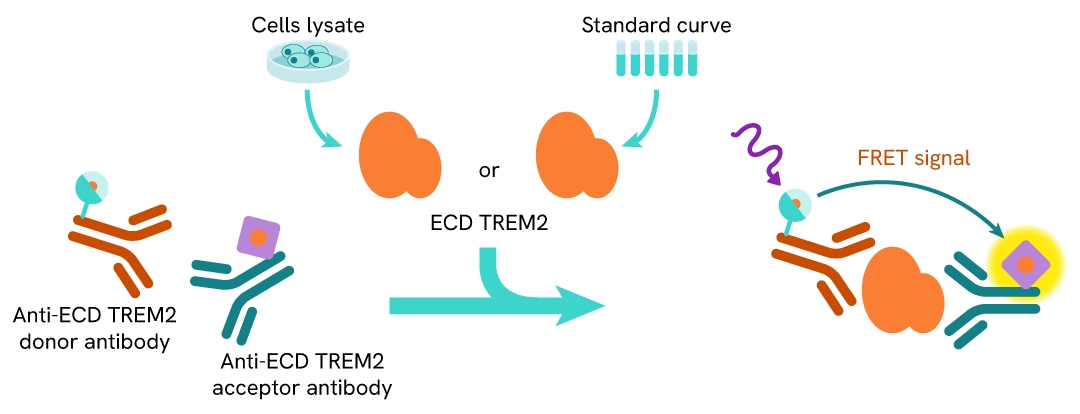
Principle of the HTRF human and mouse ectodomain TREM2 assay
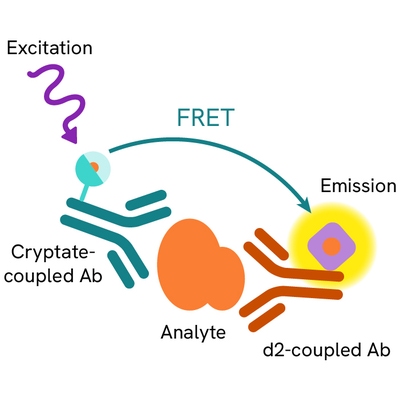
The HTRF Ectodomain TREM2 assay is based on a TR-FRET sandwich immunoassay involving two specific antibodies, one labelled with Eu3+ cryptate (donor) and the other with d2 (acceptor). Both antibodies bind to ectodomain TREM2, and the donor-acceptor proximity enables a fluorescent TR-FRET signal. The intensity of the signal is directly proportional to the concentration of ectodomain TREM2 present in the sample.

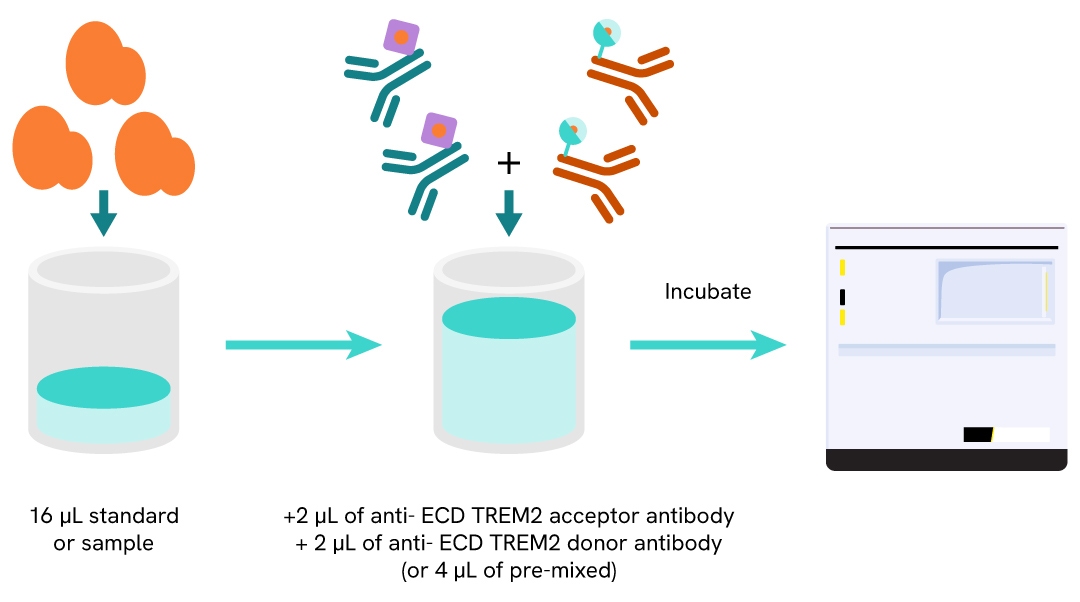
Protocol of the HTRF human and mouse ectodomain TREM2 assay
The HTRF Ectodomain TREM2 assay can be run in a 96- or 384-well low volume white detection plate (20 µL final). As described here, samples or standards are dispensed directly into the assay plate for the detection of ectodomain TREM2 by HTRF reagents. The antibodies labelled with HTRF fluorophores may be pre-mixed and added in a single dispensing step. No washing steps are needed. The protocol can be further miniaturized or upscaled by simply resizing each addition volume proportionally.
Assay details
Human and mouse ectodomain TREM2 assay details
| Sample size | 16 µL |
|---|---|
| Final assay volume | 20 µL |
| Kit component | Frozen standard, frozen detection antibodies & buffers |
| LOD & LOQ (in LB#3) | 9 & 21 pg/mL |
| Range | 81 – 12 000 pg/mL |
| Time to result | 3h at RT |
| Species | Human and Mouse |
Analytical performance
Intra-assay precision table
All of the three cell culture supernatant samples from THP-1 cells were measured 24 times, and the %CV was calculated for each sample.
| Sample | Mean [ECD-TREM2] pg/mL |
CV |
|---|---|---|
| 1 | 3121 | 2% |
| 2 | 882 | 2% |
| 3 | 317 | 3% |
| Mean CV | 2% |
Inter-assay precision table
All of the cell culture supernatant samples from THP-1 cells were measured in three independent experiments (on three different days), and the %CV was calculated for each sample.
| Sample | Mean [ECD-TREM2] pg/mL |
CV |
|---|---|---|
| 1 | 2627 | 6% |
| 2 | 1301 | 4% |
| 3 | 522 | 4% |
| Mean CV | 5% |
Dilution linearity
Samples consisted of cell culture supernatants from THP-1 cells serially diluted in RPMI 1640 complete medium. The excellent recovery percentages obtained in these experiments demonstrate the strong dilution linearity of the assay (acceptance criteria for dilution tests: 85–115%).
| Dilution factor | Expected ECD-TREM2 concentration (pg/mL) | Measured ECD-TREM2 concentration (pg/mL) | % dilution recovery |
|---|---|---|---|
| Neat | - | - | - |
| 4 | 1275 | 1103 | 86% |
| 8 | 638 | 620 | 97% |
| 16 | 319 | 323 | 101% |
| 32 | 159 | 170 | 107% |
| 64 | 80 | 90 | 112% |
| Mean CV | 101% | ||
Spike & recovery
Three known concentrations of TREM2 protein (~3 099, 542, and 164 pg/mL) were spiked into a diluted native sample from THP-1 cell supernatants. The expected concentrations were compared with the measured values to calculate antigen recovery (acceptance criteria: 80–120%). The results shown in the table below indicate that good recoveries were achieved for all three spike levels tested.
| [ECD-TREM2] protein (pg/mL) |
[ECD-TREM2] Spiked Sample (pg/mL) |
Expected [ECD-TREM2] (pg/mL) |
Measured [ECD-TREM2] (pg/mL) |
% dilution recovery |
|---|---|---|---|---|
| 164 | 655 | 819 | 758 | 92% |
| 542 | 655 | 1197 | 1081 | 90% |
| 3099 | 655 | 3754 | 3749 | 100% |
| Mean | 94% | |||
Assay validation
Inhibition of Shedding in Differentiated THP-1 Cells
THP-1 cells were seeded at 50,000 cells per well in a 96-well microplate and treated for 20 hours with 100 nM PMA at 37°C, 5% CO₂. After incubation, the cells were exposed for 48 hours to 10 µM Batimastat or GW280264X. Following treatment, the supernatant was collected, and the cells were lysed with lysis buffer #3 (1×) for 30 minutes at room temperature under gentle shaking.
Next, 16 µL of each sample were transferred into a 384-well low-volume white microplate, followed by the addition of 4 µL of premixed HTRF detection reagents. The HTRF signal was recorded after a 3-hour incubation.
As expected, the broad-spectrum MMP inhibitor Batimastat and the selective ADAM10/17 inhibitor GW280264X led to a decrease in soluble TREM2 levels, while an increase in ectodomain TREM2 was observed.
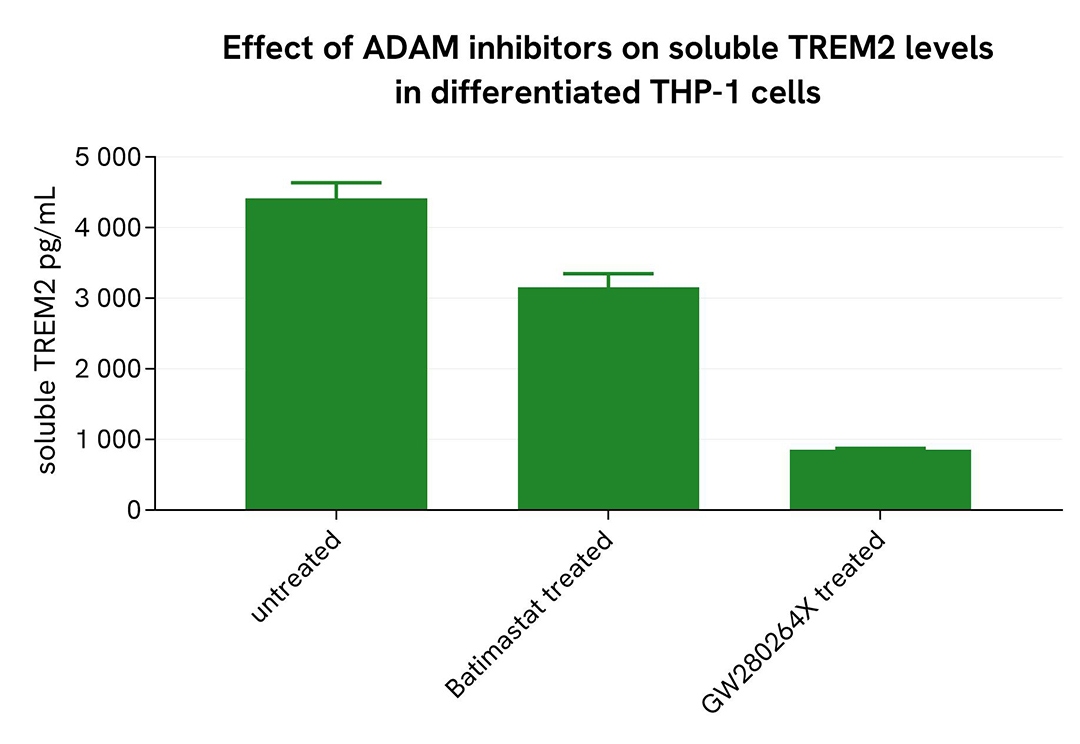
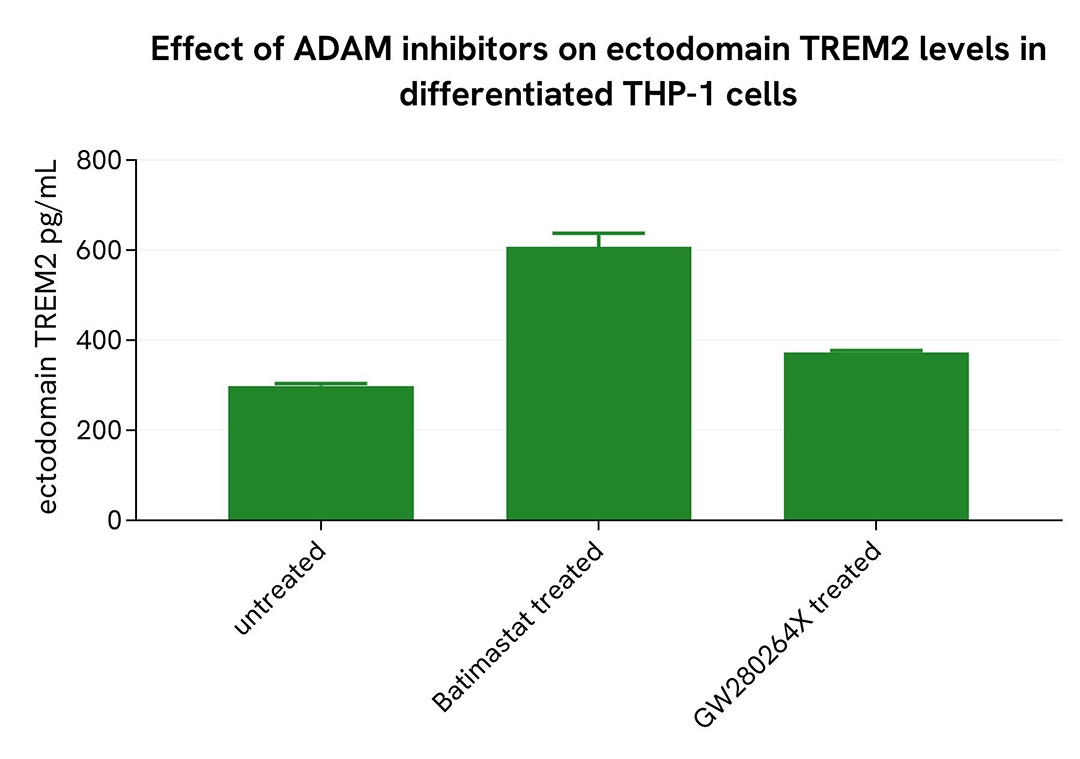
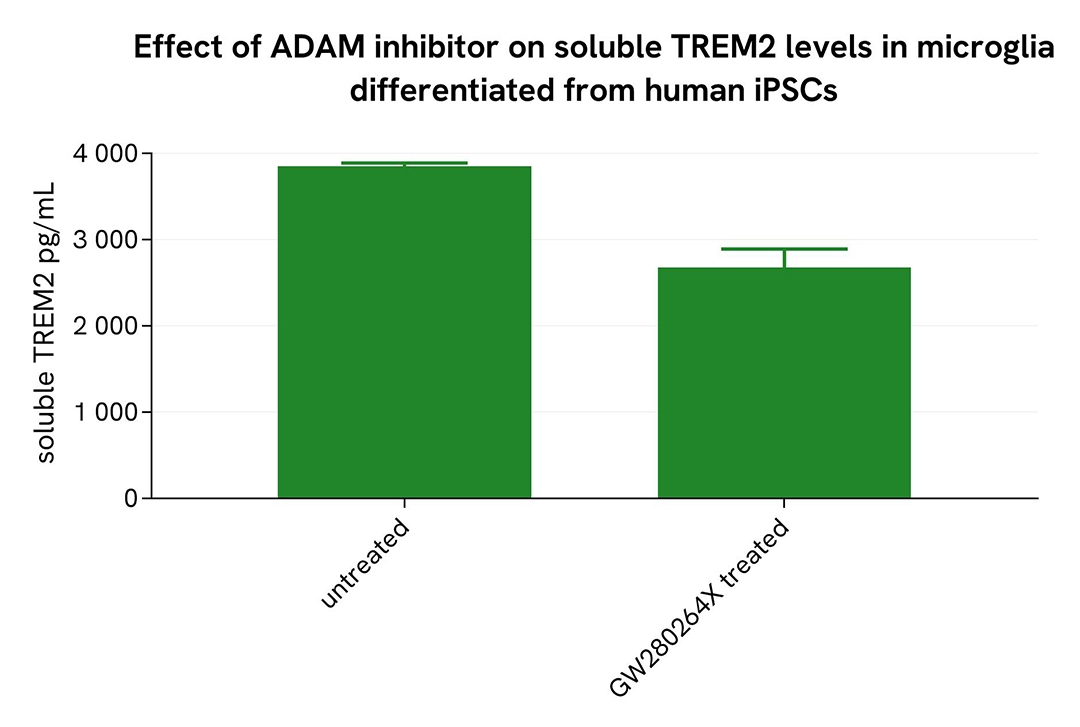
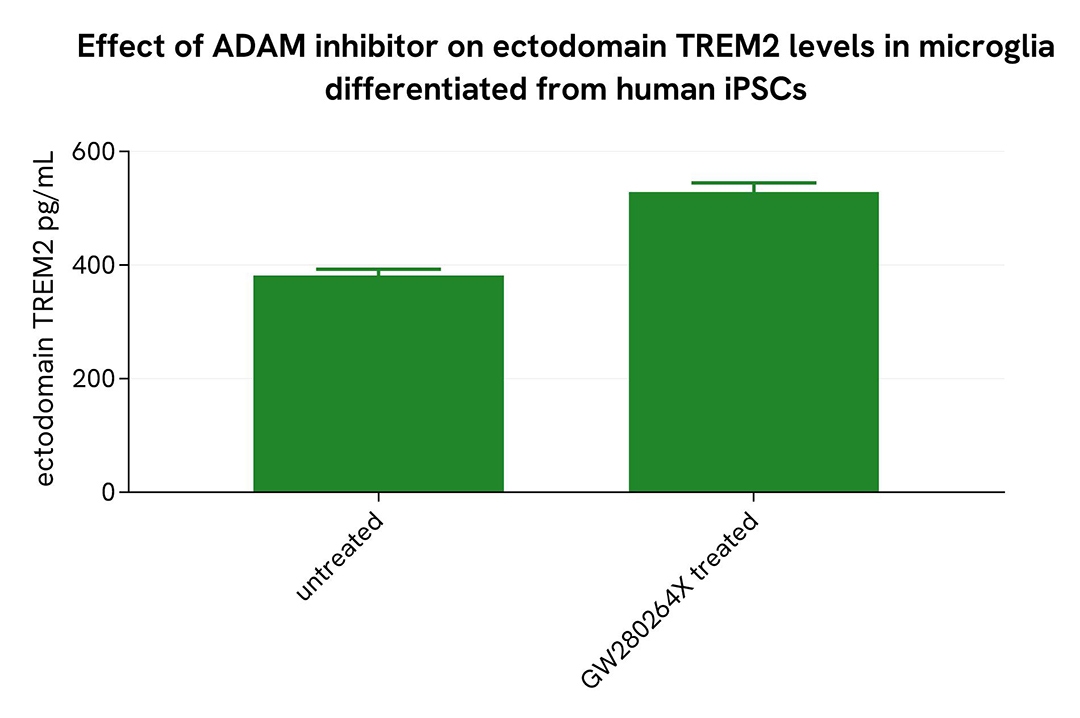
Shedding inhibition in microglia differentiated from human iPSCs
Microglia differentiated from human iPSCs (iCell® Microglia, Fujifilm Cellular Dynamics) were seeded at 6,200 cells per well in a 96-well microplate and incubated for 72 hours at 37°C, 5% CO₂. After incubation, the cells were treated for 48 hours with 10 µM GW280264X. Following centrifugation at 200×g for 3 minutes, the supernatant was collected, and the cells were lysed with lysis buffer #3 (1×) for 30 minutes at room temperature under gentle shaking.
Next, 16 µL of each sample were transferred into a 384-well low-volume white microplate, followed by the addition of 4 µL of premixed HTRF detection reagents. The HTRF signal was recorded after a 3-hour incubation.
As expected, treatment with GW280264X led to a decrease in soluble TREM2 levels, while an increase in ectodomain TREM2 was observed.
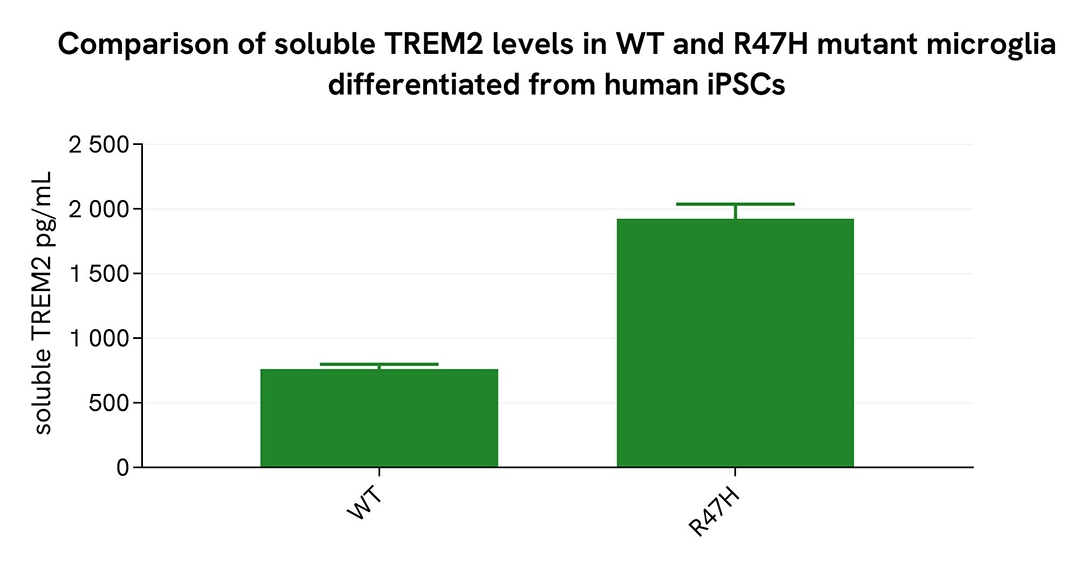
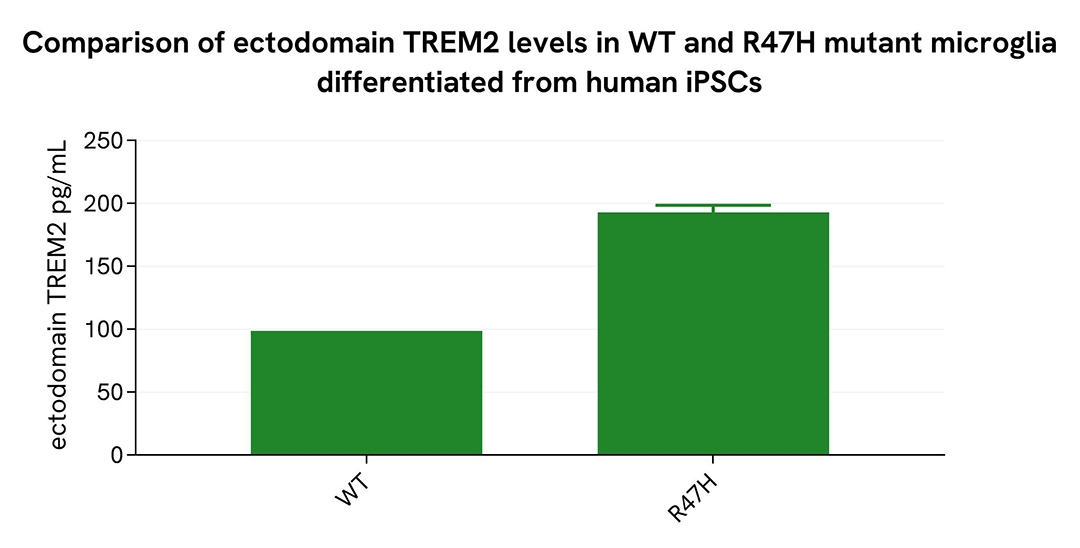
Comparison of WT and R47H Mutant Microglia Differentiated from Human iPSCs
WT and R47H microglia differentiated from human iPSCs (iCell® Microglia, Fujifilm Cellular Dynamics) were seeded at 12,500 cells per well in a 96-well microplate and incubated for 72 hours.
Following centrifugation at 200×g for 3 minutes, the supernatant was collected, and the cells were lysed with lysis buffer #3 (1×) for 30 minutes at room temperature under gentle shaking.
Next, 16 µL of each sample were transferred into a 384-well low-volume white microplate, followed by the addition of 4 µL of premixed HTRF detection reagents. The HTRF signal was recorded after a 3-hour incubation.
As expected, the R47H mutation in microglia differentiated from human iPSCs led to a significant increase in soluble and ectodomain TREM2 levels.
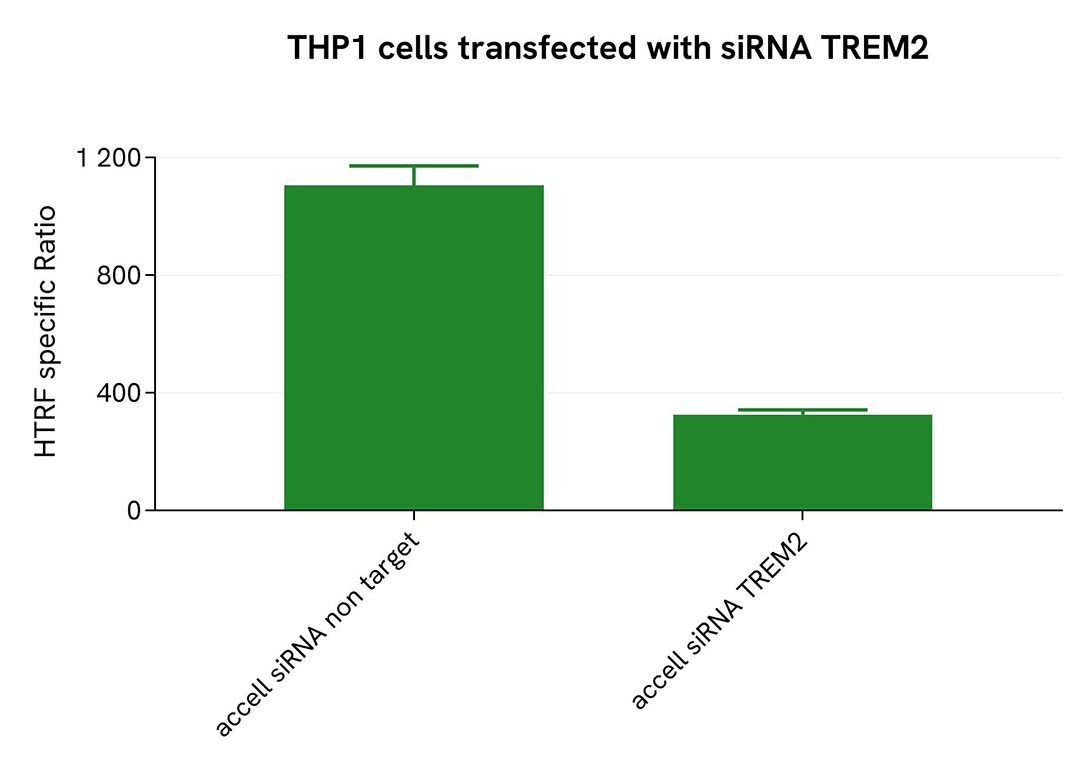
Validation of ectodomain TREM2 assay selectivity using accell siRNA
THP-1 cells were plated in a 96-well plate (25,000 cells/well) and transfected with 2.5 µM Accell siRNA targeting TREM2, along with a negative control. After a 72-hour incubation, 100 nM PMA were added to the cells for 48 hours.
For detection, 16 µL of supernatant were transferred into a 384-well low-volume white microplate, followed by the addition of 4 µL of premixed HTRF detection reagents. The HTRF signal was recorded after a 3-hour incubation.
Transfection with TREM2 siRNA resulted in a significant decrease (71%) in protein detection compared to the non-targeting control, demonstrating the selectivity of the HTRF Ectodomain TREM2 assay.
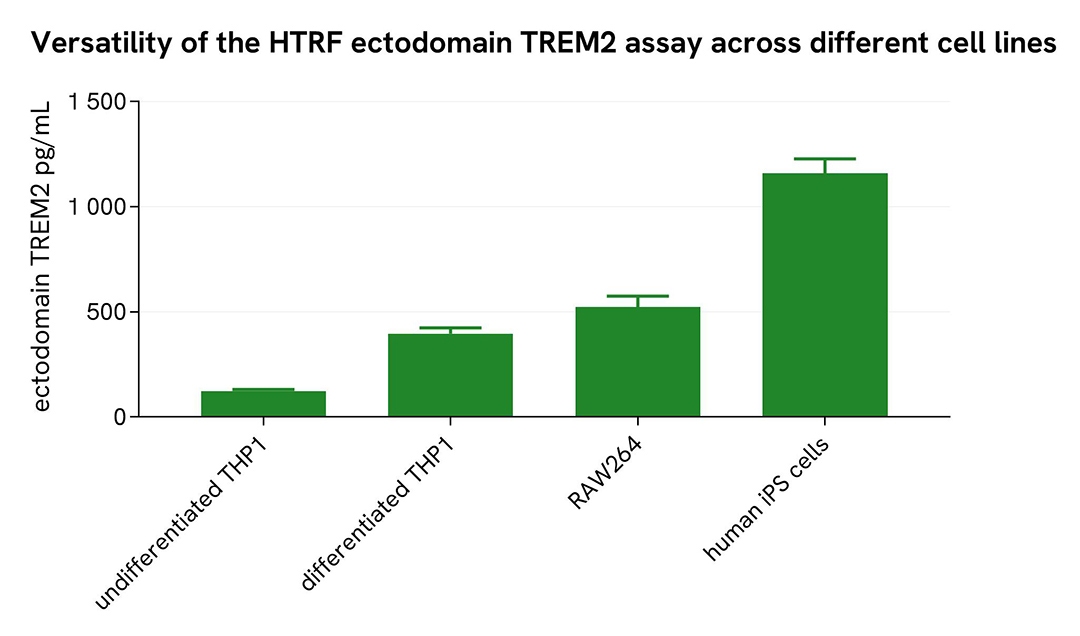
Assessment of ectodomain TREM2 levels across different cell lines
Undifferentiated and differentiated THP-1 cells (treated with 100 nM PMA for 20 hours), microglia differentiated from human iPSCs (iCell® Microglia, Fujifilm Cellular Dynamics), as well as mouse RAW264.7 cells, were seeded at 25,000 cells per well in a 96-well microplate and incubated for 20 hours. After incubation, the supernatant was collected following the protocol for adherent or suspension cells.
Next, 16 µL of each sample were transferred into a 384-well low-volume white microplate, followed by the addition of 4 µL of premixed HTRF detection reagents. The HTRF signal was recorded after a 3-hour incubation.
The HTRF ectodomain TREM2 assay successfully detected ectodomain TREM2 across various cellular models, revealing differential expression levels.
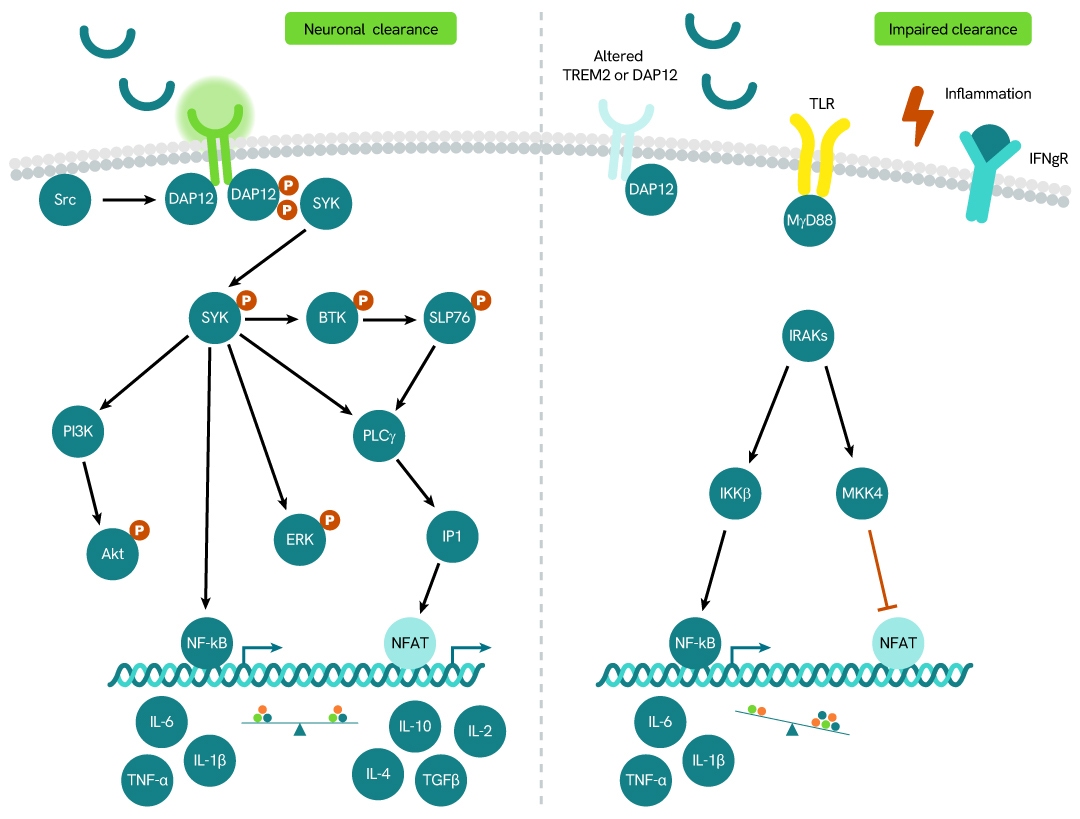
Simplified pathway
TREM2 signaling pathway
TREM2 (Triggering Receptor Expressed on Myeloid Cells 2) is a member of the TREM family of receptors, which regulate the activity of various immune cell types, including neutrophils, monocytes/macrophages, microglia, and dendritic cells. TREM2 requires the adaptor protein DAP12 for downstream signaling.
The receptor undergoes proteolytic cleavage by ADAM10 and ADAM17, a process known as shedding. This cleavage releases the ectodomain - the extracellular portion of the receptor - into the surrounding environment as soluble TREM2 (sTREM2), which has been detected in human cerebrospinal fluid (CSF). Elevated levels of sTREM2 have been observed in patients with multiple sclerosis and other inflammatory neurological conditions compared to those without inflammatory neurologic disorders.
Specifications
| Application |
Protein Quantification
|
|---|---|
| Brand |
HTRF
|
| Detection Modality |
HTRF
|
| Dynamic Range |
81 - 12,000 pg/mL
|
| Limit of Detection |
9 pg/mL
|
| Limit of Quantification |
21 pg/mL
|
| Product Group |
Kit
|
| Sample Volume |
16 µL
|
| Shipping Conditions |
Shipped in Dry Ice
|
| Target |
TREM2
|
| Target Class |
Biomarkers
|
| Target Species |
Human
Mouse
|
| Technology |
TR-FRET
|
| Therapeutic Area |
Neuroinflammation
Neuroscience
|
| Unit Size |
10,000 assay points
|
Resources
Are you looking for resources, click on the resource type to explore further.
Loading...


How can we help you?
We are here to answer your questions.






























