

HTRF AAV8 Detection Kit, 500 Assay Points
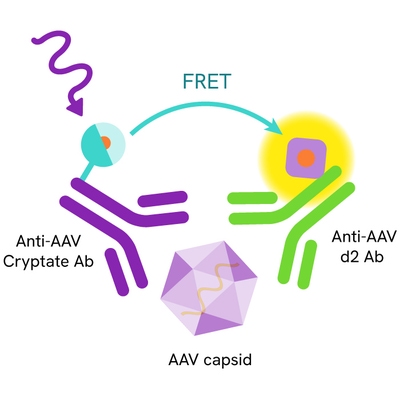



The AAV8 Capsid kit is designed for the quantitative measurement of Adeno-associated virus serotype 8 (AAV8) particles in both cell lysates and cell supernatants.
| Feature | Specification |
|---|---|
| Application | Bioprocessing |
| Sample Volume | 5 µL |
The AAV8 Capsid kit is designed for the quantitative measurement of Adeno-associated virus serotype 8 (AAV8) particles in both cell lysates and cell supernatants.






Loading...
Product information
Overview
Adeno-associated virus (AA) vectors are the leading platform for gene delivery for the treatment of a variety of human diseases. AAV Serotype 8 (AAV8) when used as vector in gene therapy, has a liver cell transduction efficiency greater than those of other serotypes which has resulted in efforts to develop this virus as a gene therapy vector for hemophilia. AAV8 also exhibits a natural tropism for other tissues such as brain, muscle and retina. The AAV8 kit is designed to detect and quantify AAV8 particles in an easy-to-use, no-wash format. The simple and robust procedure benefits from increased throughput compared to ELISA.
HTRF assays offer many advantages over other technologies:
- Homogeneous add-and-read format
- No wash steps
- Low background
- Straightforward miniaturization from 96- or 384-well microplates to high density assay formats such as 384-well low volume and 1536-well plates
- Stable signal, providing flexibility in time of readout or size of assays
How it works
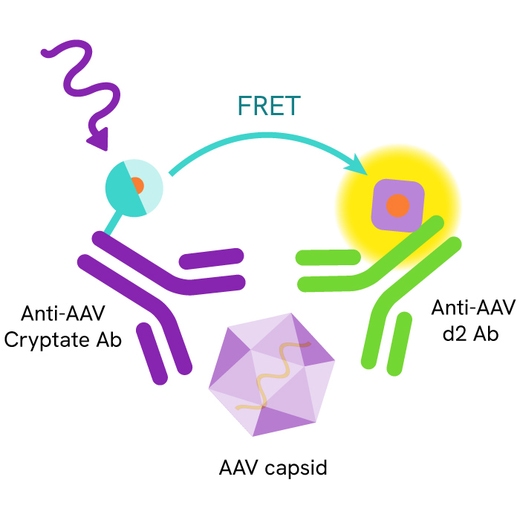
AAV8 capsid assay principle
The Adeno-Associated Virus serotype 8 (AAV8) assay measures AAV8 capsid in cell supernatant or cell lysate. The assay uses two labeled antibodies: one coupled to a donor fluorophore, the other to an acceptor. In presence of AAV8 capsid in a cell extract or supernatant the addition of these conjugates brings the donor fluorophore into close proximity with the acceptor, and thereby generates a FRET signal. Its intensity is directly proportional to the concentration of the capsid present in the sample and provides a means of assessing any changes caused by experimental variability under a no-wash assay format.

AAV8 capsid assay protocol
The AAV8 capsid assay protocol, using a 384-well small volume white plate, is described on the right. 5 µL of sample or standard and 5 µL of diluent are dispensed directly into the detection plate for detection by HTRF® reagents. The antibodies labeled with HTRF fluorophores were added before an overnight incubation. The assay can be run in up to a 1536-well format by simply resizing each addition volume proportionally.

Assay details
AAV8 capsid assay specifications

| Sample size | 5 µL |
|---|---|
| Final assay volume | 20 µL |
| Time to results | Overnight at RT |
| Detection limit (LOD) in diluent | 2.50E+08 VP/mL |
| Dynamic Range | 3.09E+09 – 2.50E+11 VP/mL |
| Sample compatibility |
From raw harvest material to the final product Supernatant, Cell Lysate (LB#3) |
Analytical performance
Precision
Intra-assay
| Sample | Mean [AAV8] (VP/mL) | CV |
|---|---|---|
| 1 | 1.60E+11 | 3% |
| 2 | 1.00E+10 | 4% |
| 3 | 5.00E+10 | 5% |
| Mean CV | 4% |
Each of the 3 samples was measured 24 times, and % CV was calculated for each sample.
Inter-assay
| Sample | Mean [AAV8] (VP/mL) | CV |
|---|---|---|
| 1 | 2.00E+11 | 1% |
| 2 | 1.00E+11 | 7% |
| 3 | 5.00E+10 | 8% |
| Mean CV | 5% |
Each of the samples was measured in 3 independent experiments performed by different operators, and % CV was calculated for each sample.
Dilutional linearity
The excellent % of recovery obtained from these experiments shows the good dilution linearity of the assay. Samples were AAV8 empty capsids serially diluted with the kit DIL5 diluent.
|
Dilution Factor |
[AAV8] Expected (VP/mL) |
[AAV8] Measured (VP/mL) |
Dilution Recovery |
|---|---|---|---|
|
Neat |
- |
2.00E+11 |
100% |
|
2 |
1.05E+11 |
1.03E+11 |
98% |
|
4 |
5.26E+10 |
4.82E+10 |
92% |
|
8 |
2.63E+10 |
2.31E+10 |
88% |
|
16 |
1.31E+10 |
1.21E+10 |
|
|
Mean |
94% |
Interferences
Different cell lines (SF9 and HEK293), representative of AAV manufacturing processes, were seeded in T175 flasks in complete culture medium at 37°C, 5% CO2. The cells were then lysed with 3 mL of lysis buffer #3 (1X) for 30 minutes at RT under gentle shaking.
AAV8 capsid at 6.0E+10 VP/mL was spiked in several dilutions of SF9 or HEK293 cell lysates not expressing AAV capsids (corresponding to different amount of total protein concentration, mg/mL). All samples, including lysates without capsid added were measured in the assay.
As expected, no AAV8 was detected in SF9 or HEK293 control cell lysates. However, for an accurate quantification of AAV8 concentration it is important to ensure that total protein concentration is below 1 mg/mL for SF9 cell lysates and HEK293 cell lysates by using dilution with lysis buffer #3.
At higher cell lysate total protein concentration, there is an interference that could affect the accuracy of the AAV8 concentration measurement.
|
[AAV8] (VP/mL) |
[Total Protein] SF9 cell lysates Spiked Sample (mg/mL) |
Recovery |
|---|---|---|
| 6.0E+10 | 1 | 101% |
| 0.5 | 107% | |
| 0.25 | 105% | |
| 0.125 | 160% | |
| 0.05 | 102% |
|
[AAV8] (VP/mL) |
[Total Protein] HEK293 cell lysates Spiked Sample (mg/mL) |
Recovery |
|---|---|---|
| 6.0E+10 | 1 | 109% |
| 0.5 | 107% | |
| 0.25 | 102% | |
| 0.125 | 107% | |
| 0.05 | 111% |
Antigen spike and recovery
Three different concentrations of AAV8 capsid were independently mixed with SF9 or HEK293 cell lysates diluted in lysis buffer #3 at 1 mg/ml. The measured concentrations were compared to the expected values and expressed as percentage of recovery.
|
Sample |
[AAV8] Standard (VP/mL) |
|
|---|---|---|
|
Recovery |
||
|
1 |
1,34E+10 |
116% |
|
2 |
2,93E+10 |
108% |
|
3 |
5,41E+10 |
115% |
|
|
|
113% |
|
Sample |
[AAV8] Standard (VP/mL) |
|
|---|---|---|
|
Recovery |
||
|
1 |
1,34E+10 |
118% |
|
2 |
2,93E+10 |
104% |
|
3 |
5,41E+10 |
116% |
|
|
|
113% |
Cross reactivities
Cross reactivities were assessed using other serotypes from the AAV family. Standard curves were generated for each serotype using AAV capsids diluted in the kit diluent.
5 µL of capsids were transferred into a white detection plate (384 low volume), followed by 5 µL of diluent then 10 µL of the HTRF AAV8 capsid detection reagents. The HTRF signal was recorded after an overnight incubation at room temperature.
Signals were interpolated on the assay standard curve to interpolate concentrations. The assay is specific to AAV8 as other serotypes were not detected using this assay.
|
Empty AAVs Particles |
Cross Reactivity |
|---|---|
|
AAV8 |
100% |
|
AAV1 |
No |
|
AAV2 |
No |
|
AAV3B |
No |
|
AAV5 |
No |
|
AAV6 |
No |
|
AAV9 |
No |
Assay validation
Validation of HTRF AAV8 capsid detection on full and empty AAV8 particles
To demonstrate the detection of both full and empty AAV8 capsids, recognition of full AAV8-CMV-eGFP and empty AAV8 capsids was analyzed in the assay. A large range of AAV8-CMV-eGFP concentration (GC/mL) was calculated in VP/mL using an independent sample quantitation assay.
5 µL of full or empty capsids diluted in the kit diluent were then transferred into a white detection plate (384 low volume), followed by 5 µL of diluent and 10 µL of the HTRF AAV8 capsid detection reagents. The HTRF signal was recorded after an overnight incubation at room temperature.
As expected, the HTRF AAV8 capsid detection assay can detect in the same way both full and empty AAV8 capsids.

Specifications
| Application |
Bioprocessing
|
|---|---|
| Brand |
HTRF
|
| Detection Modality |
HTRF
|
| Lysis Buffer Compatibility |
Lysis Buffer 1
Lysis Buffer 2
Lysis Buffer 3
Lysis Buffer 4
|
| Product Group |
Kit
|
| Sample Volume |
5 µL
|
| Shipping Conditions |
Shipped in Dry Ice
|
| Target Class |
Viral Particles
|
| Technology |
TR-FRET
|
| Unit Size |
500 assay points
|
Video gallery
Resources
Are you looking for resources, click on the resource type to explore further.
What Are the Challenges and Solutions?
All across the globe, AAVs are getting the attention of scientists and companies working in...
Discover the versatility and precision of Homogeneous Time-Resolved Fluorescence (HTRF) technology. Our HTRF portfolio offers a...
This guide provides you an overview of HTRF applications in several therapeutic areas.
Neurodegenerative diseases, such as Alzheimer’s, Parkinson’s, and Huntington’s, are complex disorders that affect millions...
See how researchers boost their gene therapy studies with Revvity technologies
The absence or mutation of protein is a factor that...
Loading...


How can we help you?
We are here to answer your questions.






























