
Development of novel AAV capsids for gene therapy
Revvity Gene Delivery, where SIRION Biotech’s expertise lives on, has been supporting pharmaceutical and biotech organizations for years enabling targeted gene therapy by providing our IP and know-how in AAV capsid engineering, creating new intellectual property (IP) tailored specifically to our partner’s needs.
AAV capsid directed evolution
To optimize AAV tropism, we use a directed evolution approach via capsid randomization. This technology is made available through the partnership and license between SIRION Biotech and Prof. Dr. Dirk Grimm of Heidelberg University in Germany, a pioneer in AAV capsid evolution.
AAV directed capsid evolution entails:
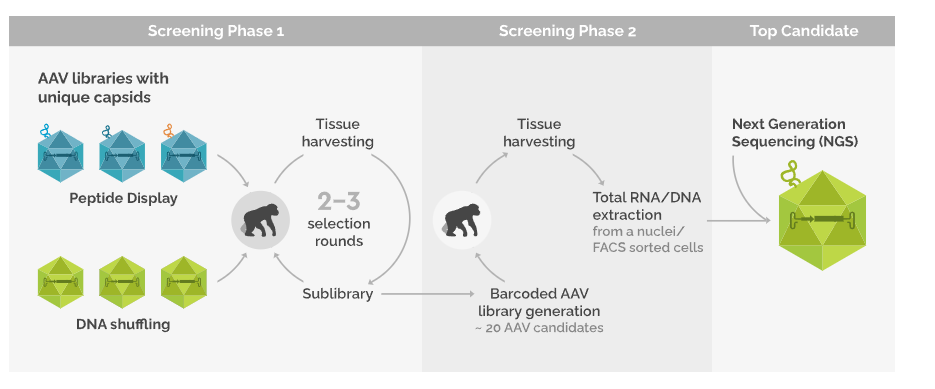
- Iterative in vivo screenings of recombinant AAV (rAAV) libraries harboring randomized peptide insertion libraries or AAV shuffled variants or combinations thereof
- In vivo biodistribution analysis of enriched barcoded AAVs.
- AAV lead candidates are identified by bioinformatic analyses of the data collections obtained from state-of-the-art next generation sequencing and PCR techniques from transduced tissue.
AAV rational design – nanobody targeting
Highly specific nanobodies are the basis of a novel rational design strategy that allows direct targeting of a surface protein of interest. This technology can be implemented separately or in combination with directed evolution of AAV capsids to further enhance cell or tissue tropism and minimize off-target effects.
Questions?
We’re here to help.
Contact us Please note that product labeling (such as kit insert, product label, and kit box) may be different compared to the company branding. Please contact your local representative for further details.




























