
HTRF Human and Mouse Phospho-CDC6 (Ser54) Detection Kit, 10,000 Assay Points

This HTRF kit allows for the cell-based quantitative detection of CDC6 when phosphorylated at Ser54.
| Feature | Specification |
|---|---|
| Application | Cell Signaling |
| Sample Volume | 16 µL |
This HTRF kit allows for the cell-based quantitative detection of CDC6 when phosphorylated at Ser54.



Product information
Overview
CDC6, or Cell Division Cycle 6, is a member of the AAA+ ATPase family and plays a crucial role in the regulation of DNA replication. It is an essential component of the pre-replication complex, facilitating its assembly at origins of replication to ensure that DNA replication occurs only once per cell cycle. Beyond its role in replication initiation, CDC6 also contributes to the activation and maintenance of cell cycle checkpoints, which are vital for preserving genomic stability.
HTRF assays offer many advantages over other technologies:
- Homogeneous add-and-read format
- No wash steps
- Low background
- Straightforward miniaturization from 96- or 384-well microplates to high density assay formats such as 384-well low volume and 1536-well plates
- Stable signal, providing flexibility in time of readout or size of assays
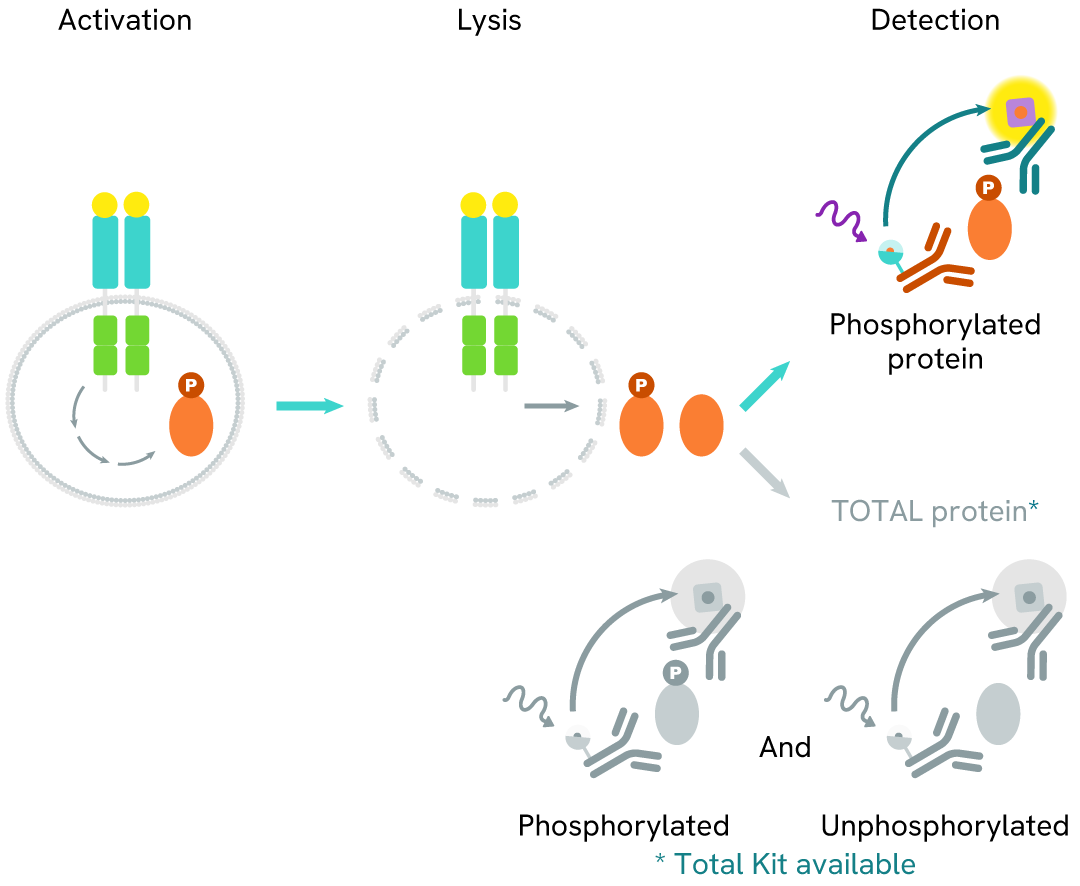
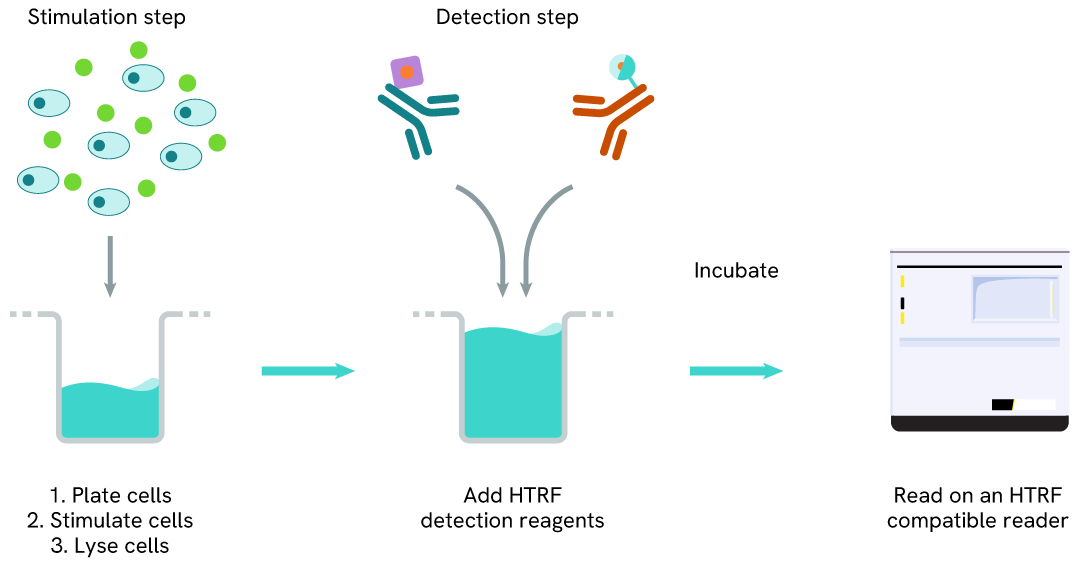
How it works
Phospho-CDC6 (Ser54) assay principle
The Phospho-CDC6 (Ser54) assay measures CDC6 phosphorylated at Ser54. Unlike Western Blot, the assay is entirely plate-based and does not require gels, electrophoresis, or transfer.
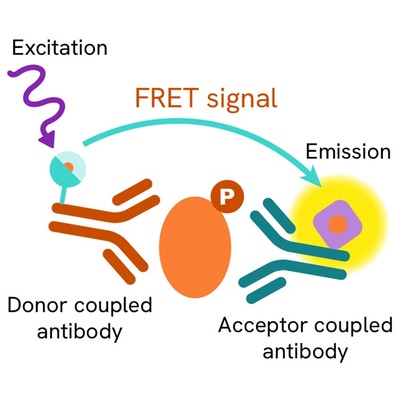
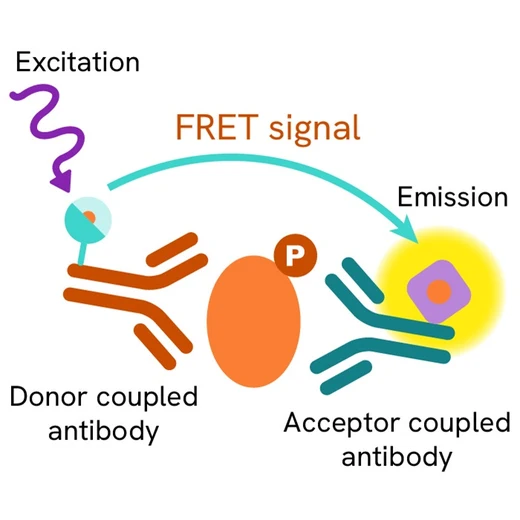
The assay uses two antibodies: one labeled with a donor fluorophore, and the other with an acceptor fluorophore. The first antibody was selected for its specific binding to the phosphorylated motif on the protein, while the second recognizes the protein independently of its phosphorylation state.
Phosphorylation of the protein enables the formation of an immune complex involving both labeled antibodies, bringing the donor fluorophore into close proximity to the acceptor. This interaction generates a FRET signal, whose intensity is directly proportional to the concentration of phosphorylated protein in the sample. The assay provides a reliable means of assessing the protein's phosphorylation state in a no-wash format.

Phospho-CDC6 (Ser54) two-plate assay protocol
The two-plate protocol involves culturing cells in a 96-well plate before lysis, then transferring lysates into a 384-well low volume detection plate before the addition of Phospho-CDC6 (Ser54) HTRF detection reagents. This protocol allows the cells' viability and confluence to be monitored.
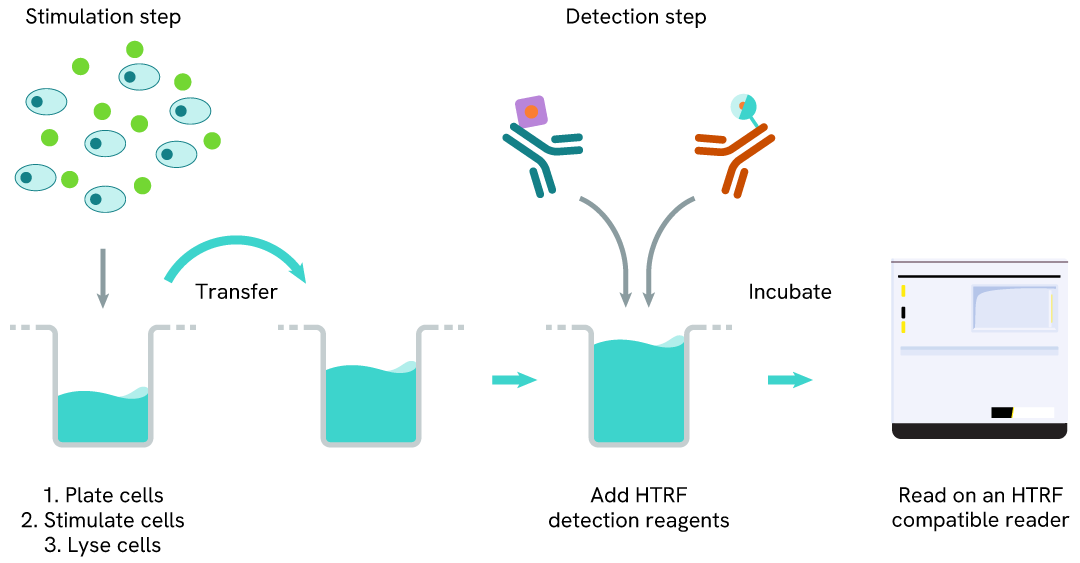
Phospho-CDC6 (Ser54) one-plate assay protocol
Detection of Phosphorylated CDC6 (Ser54) with HTRF reagents can be performed in a single plate used for culturing, stimulation, and lysis. No washing steps are required. This HTS designed protocol allows miniaturization while maintaining HTRF quality.
Assay validation
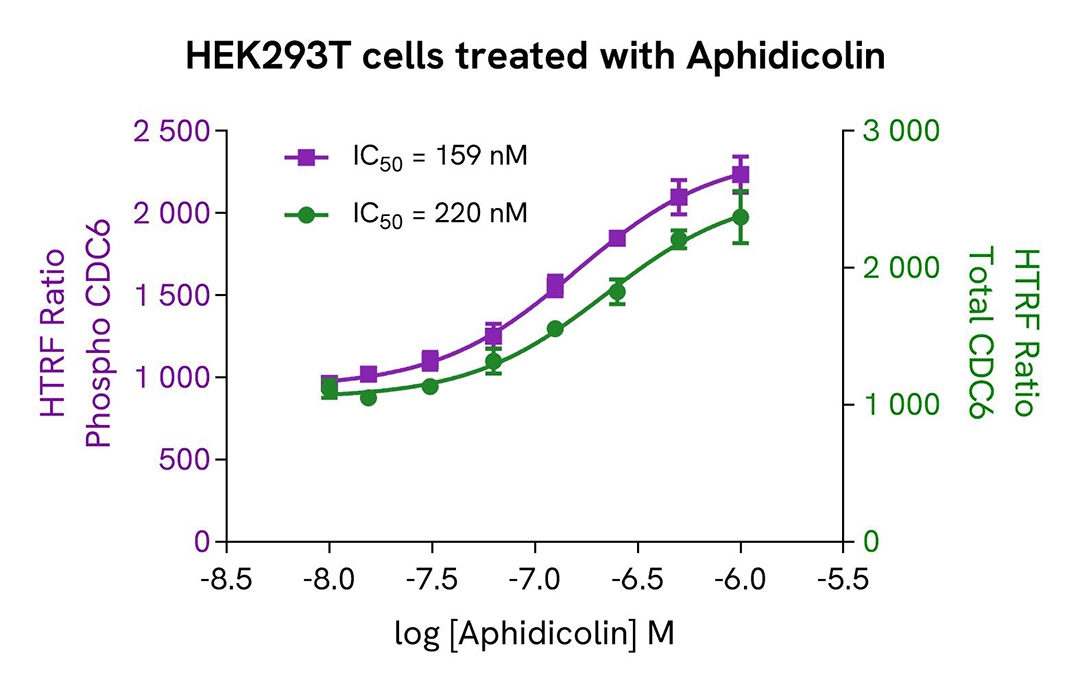
Induction of Phospho-CDC6 (Ser54) in HEK293T cells
HEK293T cells were seeded in a 96-well culture-treated plate at 25,000 cells per well in complete culture medium, and incubated overnight at 37°C with 5% CO₂. The cells were then treated for 20 hours with increasing concentrations of Aphidicolin.
After treatment, the cells were lysed with 50 µL of supplemented lysis buffer #1 for 30 minutes at room temperature under gentle shaking, following the protocol for adherent cells.
For the detection step, 16 µL of cell lysate were transferred into a 384-well low-volume white microplate, and 4 µL of HTRF Phospho-CDC6 (Ser54) or Total CDC6 detection reagents were added. The HTRF signal was recorded after overnight incubation.
As expected, the DNA-dependent polymerase inhibitor Aphidicolin induced a dose-dependent increase in the levels of Phospho-CDC6 (Ser54) and Total CDC6.
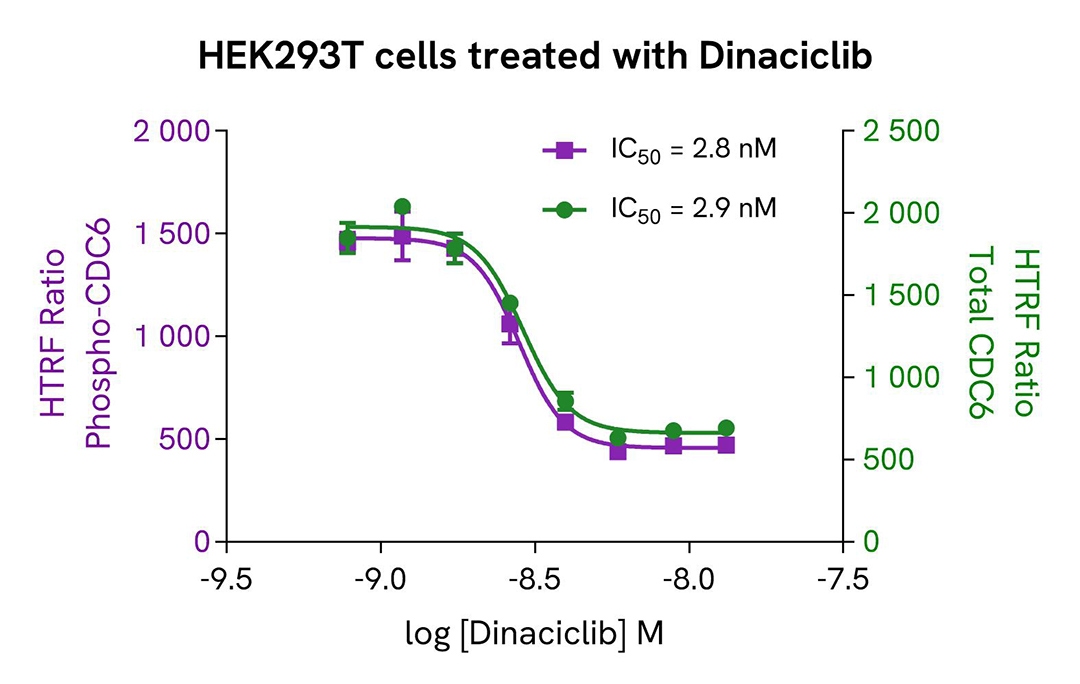
Inhibition of Phospho-CDC6 (Ser54) in HEK293T cells
HEK293T cells were seeded in a 96-well culture-treated plate at 100,000 cells per well in complete culture medium and incubated overnight at 37°C with 5% CO₂. The cells were then treated for 20 hours with increasing concentrations of Dinaciclib.
After treatment, the cells were lysed with 50 µL of supplemented lysis buffer #1 for 30 minutes at room temperature under gentle shaking, following the protocol for adherent cells.
For the detection step, 16 µL of cell lysate were transferred into a 384-well low-volume white microplate, and 4 µL of HTRF Phospho-CDC6 (Ser54) or Total CDC6 detection reagents were added. The HTRF signal was recorded after overnight incubation.
As expected, the direct CDK2 inhibitor Dinaciclib induced a dose-dependent decrease in the levels of Phospho-CDC6 (Ser54) and Total CDC6.
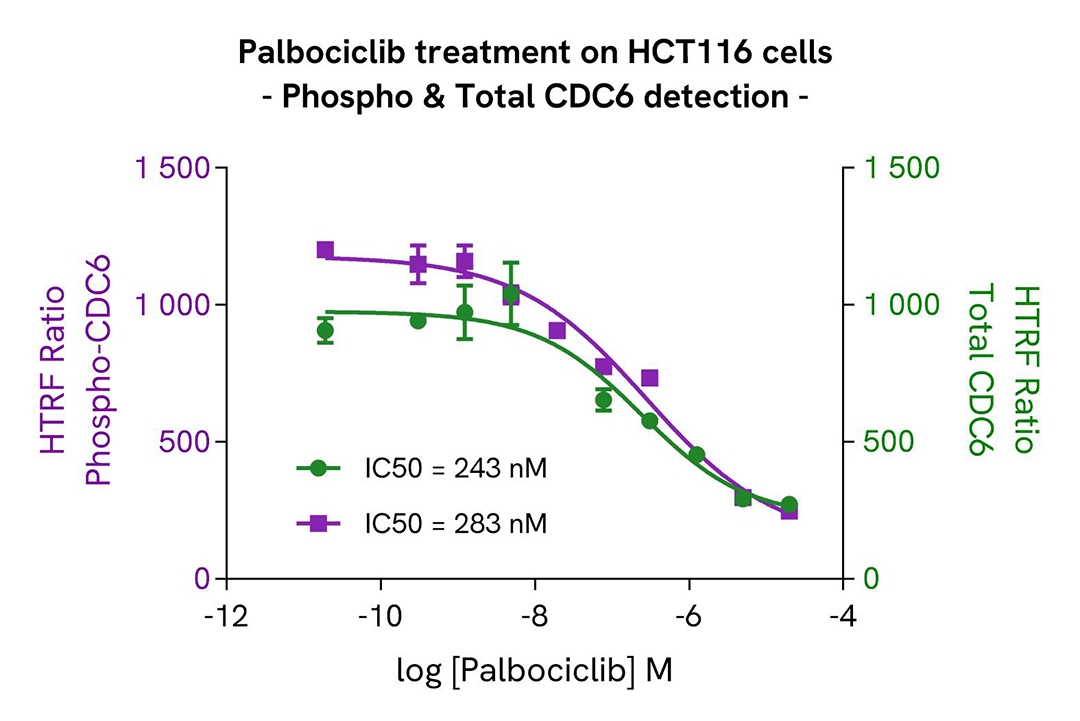
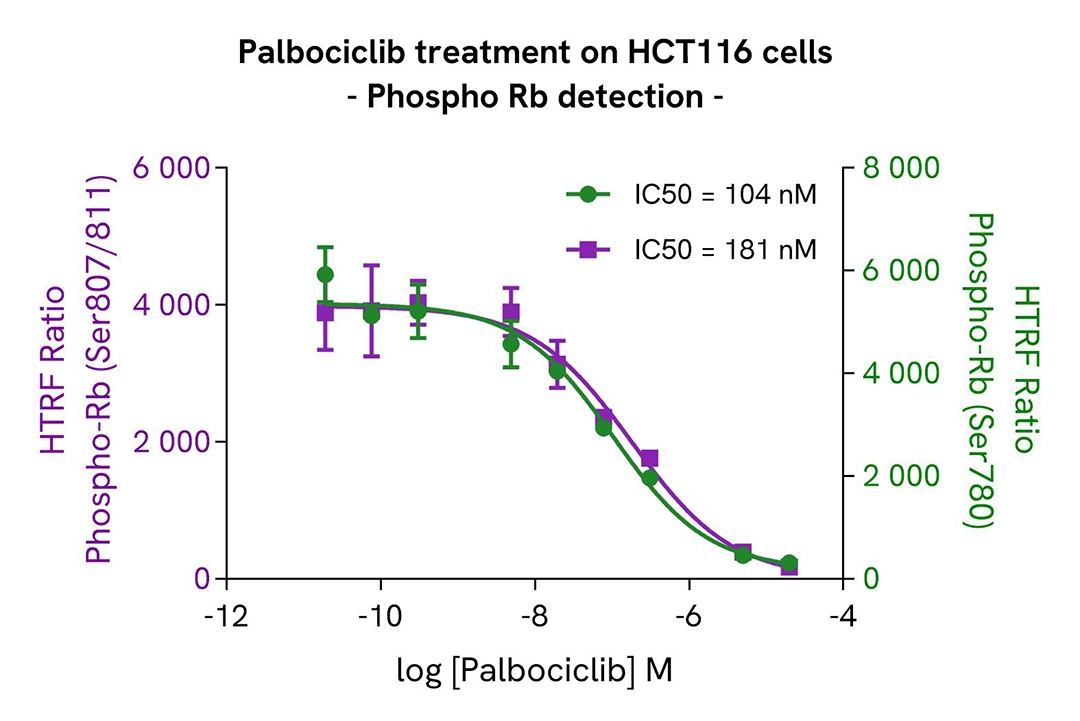
Palbociclib-Induced modulation of CDC6 and Rb phosphorylation
HCT116 cells were seeded in a 96-well culture-treated plate at 20,000 cells per well in complete culture medium and incubated at 37°C with 5% CO₂. After 6 hours, the cells were treated for 48 hours with increasing concentrations of Palbociclib.
Following treatment, the cells were lysed with 50 µL of supplemented lysis buffer #1 for 30 minutes at room temperature under gentle shaking, according to the protocol for adherent cells.
For the detection step, 16 µL of cell lysate were transferred into a 384-well low-volume white microplate, and 4 µL of premixed HTRF detection reagents were added. The HTRF signal was recorded after overnight incubation.
As expected, the Cyclin-Dependent Kinase inhibitor Palbociclib led to a significant decrease in the phosphorylation of Rb at Serine 780 and 807/811, accompanied by a dose-dependent reduction in the levels of Phospho-CDC6 (Ser54) and Total CDC6.
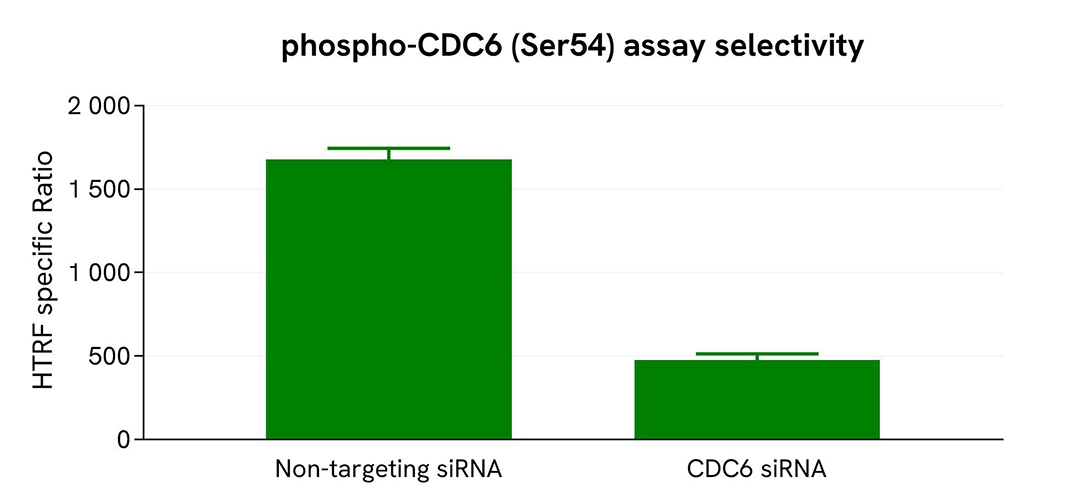
Validation of Phospho-CDC6 (Ser54) assay selectivity using ON-TARGETplus siRNA
HEK293T cells were plated in a 96-well plate at 12,500 cells per well and transfected with 50 nM ON-TARGETplus siRNA targeting CDC6, along with a non-targeting negative control. Following a 48-hour incubation, the cells were lysed with supplemented lysis buffer #1 (4×) for 30 minutes at room temperature under gentle shaking.
For the detection step, 16 µL of lysate were transferred into a 384-well low-volume white microplate, and 4 µL of HTRF Phospho-CDC6 (Ser54) detection antibodies were added. The HTRF signal was recorded after 24 hours.
Transfection with CDC6 siRNA resulted in a significant 74% decrease in protein detection compared to the non-targeting control, demonstrating the selectivity of the HTRF Phospho-CDC6 (Ser54) assay.
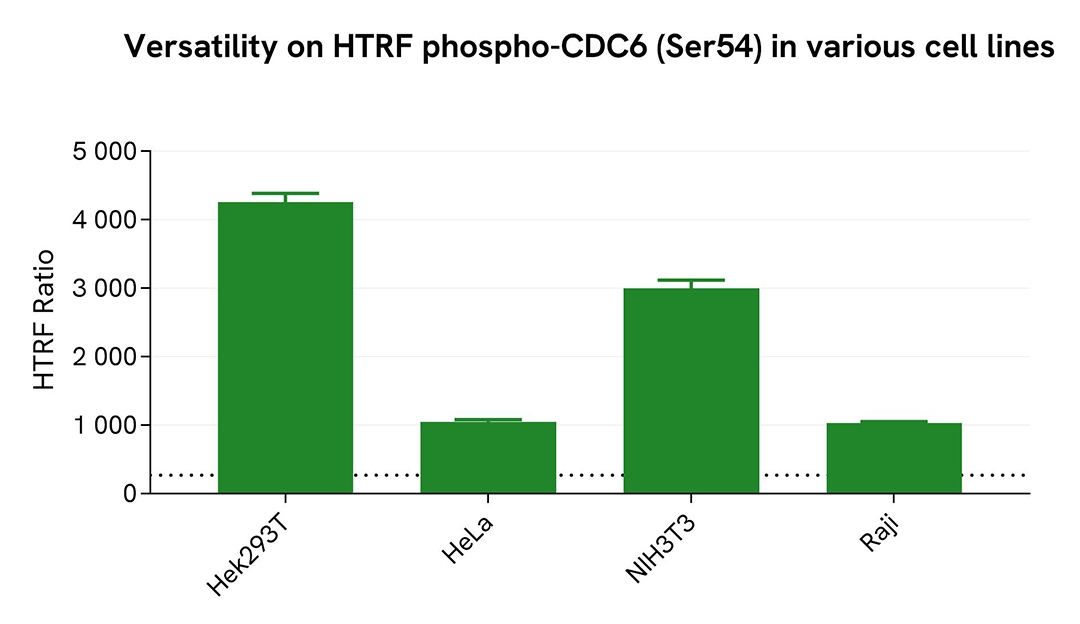
Assessment of Phospho-CDC6 (Ser54) levels in various cell lines
Adherent human cells HeLa (cervical), HEK293T (kidney) and suspension cells Raji (lymphoblast-like), as well as mouse fibroblast cells (NIH-3T3), were seeded at 100,000 cells per well in a 96-well microplate and treated for 20 hours with 500nM of Aphidicolin. After incubation, the cells were lysed for 30 minutes with supplemented lysis buffer #1 following the protocol for adherent or suspension cells, at room temperature under gentle shaking.
16 µL of lysate were transferred into a 384-well low-volume white microplate, followed by the addition of 4 µL of HTRF Phospho-CDC6 (Ser54) detection reagents. The HTRF signal was recorded after overnight incubation.
The HTRF Phospho-CDC6 (Ser54) assay successfully detected CDC6 phosphorylation at Ser54 across various cellular models, revealing different phosphorylation levels.
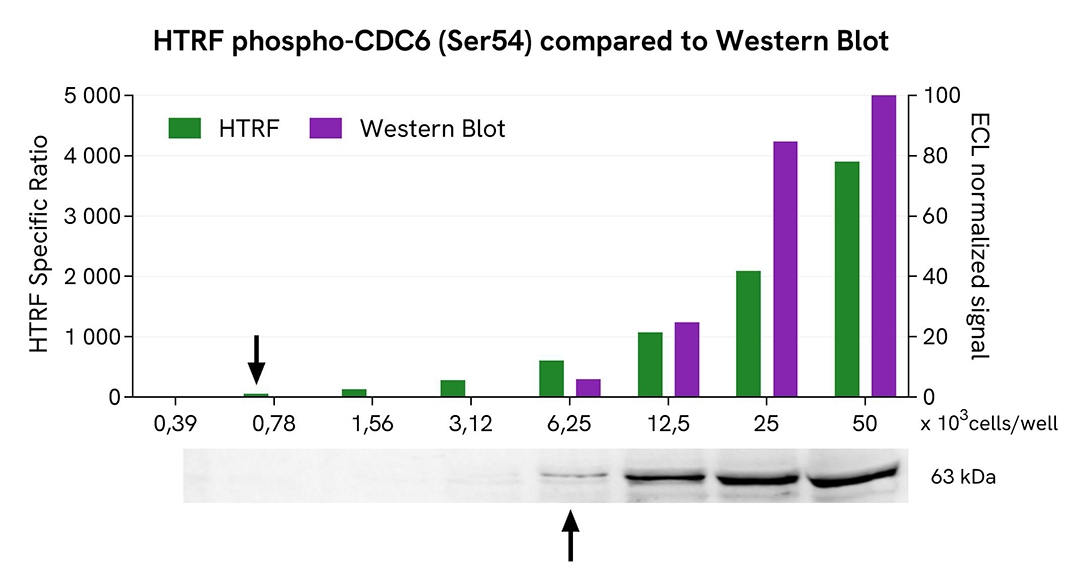
HTRF Phospho-CDC6 (Ser54) assay compared to Western Blot
HEK293T cells were grown in a T175 flask in complete culture medium at 37°C and 5% CO2 until they reached 80% confluence. The cells were then treated with 500nM of Aphidicolin for 20h and lysed with 3 mL of supplemented lysis buffer #1 (4x) for 30 minutes at room temperature under gentle shaking.
Serial dilutions of the cell lysate were performed using supplemented lysis buffer. Then 13 µL of each dilution were transferred into a low volume white microplate, followed by the addition of 3 µL of supplemented lysis buffer #1 (1x) to ensure the same quantity in both techniques. Next, 4 µL of HTRF Phospho-CDC6 (Ser54) detection reagents were added. Equal amounts of lysates were used for a side-by-side comparison between HTRF and Western Blot.
Using the HTRF Phospho-CDC6 (Ser54) assay, 780 cells/well were sufficient to detect a significant signal, while 6250 cells were needed to obtain a minimal chemiluminescent signal using Western Blot. Therefore, under these conditions, the HTRF Phospho-CDC6 (Ser54) assay was 8 times more sensitive than the Western Blot technique.
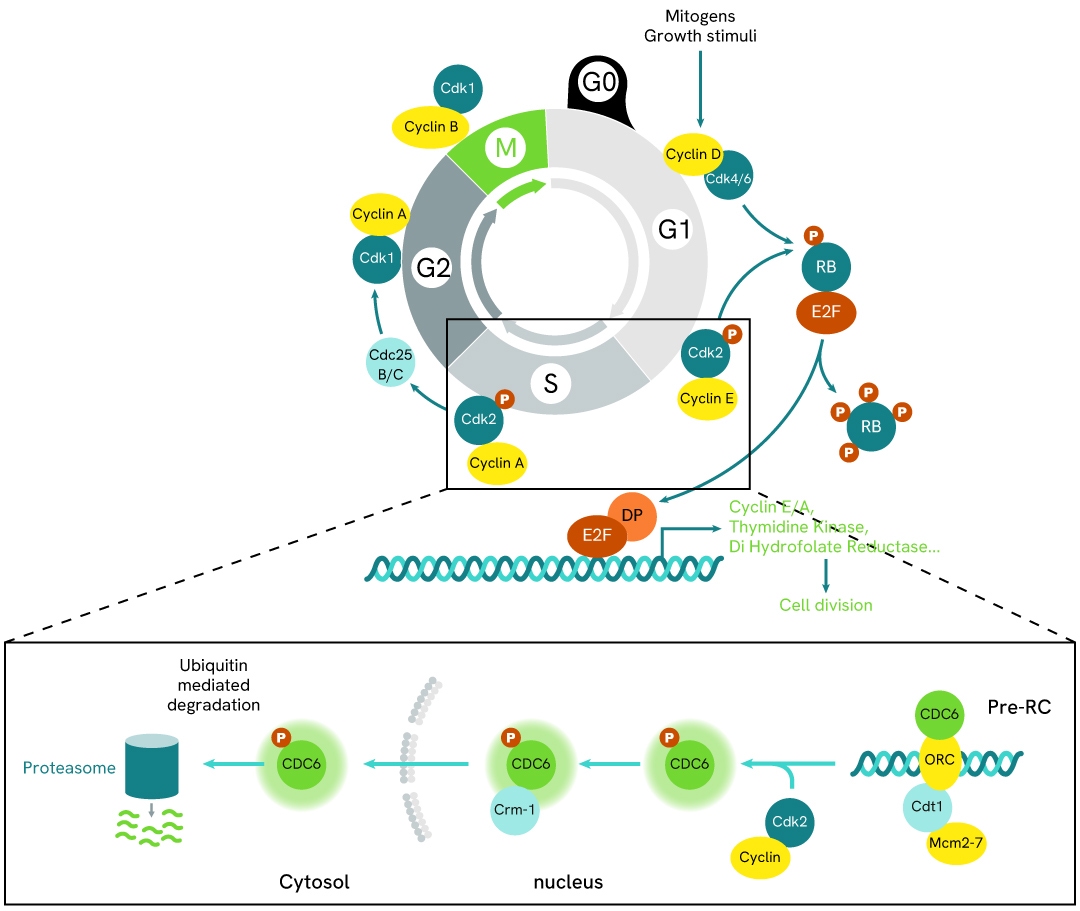
Simplified pathway
CDC6 signaling pathway
During the G1 phase, CDC6 is unphosphorylated and localized in the nucleus. It is recruited to replication origins by the origin recognition complex (ORC) in association with Cdt1. Subsequently, the Mcm2-7 complex is loaded onto the origin to form the pre-replicative complex (pre-RC). This complex renders the genome competent for replication; however, initiation only occurs after activation by cyclin-dependent kinases (CDKs) at the onset of the S phase.
At the G1/S transition, CDC6 is specifically phosphorylated by cyclin E/Cdk2 and cyclin A/Cdk2 at replication origins, promoting its dissociation from the pre-RC. CDKs also facilitate the loading of additional proteins required for DNA replication initiation onto chromatin, including Cdc45, replication protein A, and DNA polymerase.
Once DNA replication begins, phosphorylated CDC6 is exported from the nucleus to the cytoplasm via Exportin 1 (Crm1)-dependent protein export.
CDC6 is regulated by several E3 ubiquitin ligase complexes under different conditions, including APC/C-CDH1, SCF-CDC4, CRL4-Cdt2, and SCF-Cyclin F.
Specifications
| Application |
Cell Signaling
|
|---|---|
| Brand |
HTRF
|
| Detection Modality |
HTRF
|
| Lysis Buffer Compatibility |
Lysis Buffer 1
|
| Molecular Modification |
Phosphorylation
|
| Product Group |
Kit
|
| Sample Volume |
16 µL
|
| Shipping Conditions |
Shipped in Dry Ice
|
| Target |
CDC6
|
| Target Class |
Phosphoproteins
|
| Target Species |
Human
Mouse
|
| Technology |
TR-FRET
|
| Therapeutic Area |
Oncology
|
| Unit Size |
10,000 assay points
|
Resources
Are you looking for resources, click on the resource type to explore further.


Loading...
How can we help you?
We are here to answer your questions.






























