
AlphaLISA SureFire Ultra Human Total Myd88 Detection Kit, 10,000 Assay Points

 View All
View All
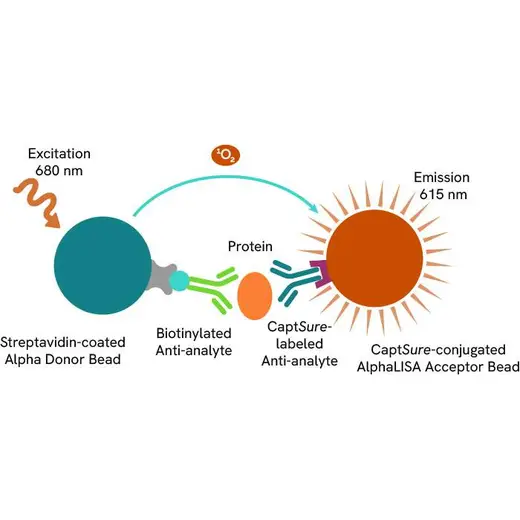



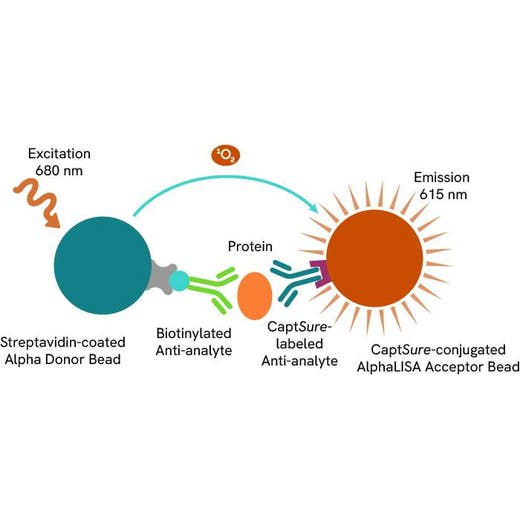



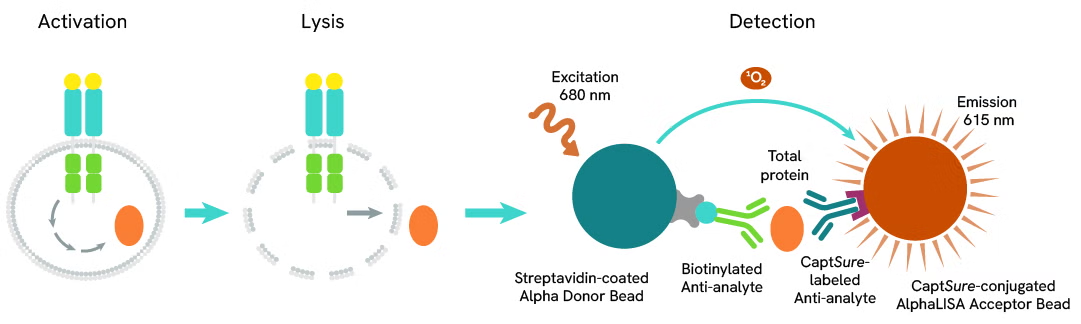
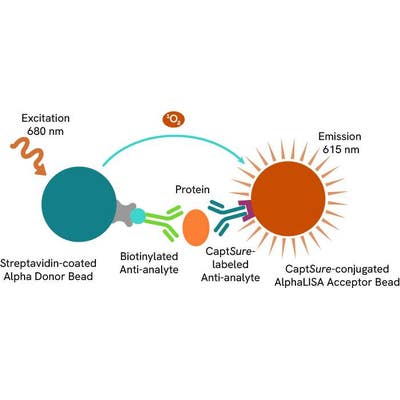
The AlphaLISA™ SureFire® Ultra™ Human Total MYD88 assay is a sandwich immunoassay for quantitative detection of total MYD88 in cellular lysates using Alpha technology.
| Feature | Specification |
|---|---|
| Application | Cell Signaling |
| Sample Volume | 10 µL |
The AlphaLISA™ SureFire® Ultra™ Human Total MYD88 assay is a sandwich immunoassay for quantitative detection of total MYD88 in cellular lysates using Alpha technology.












Loading...
Product information
Overview
Toll-like or interleukin-1 receptors (TLR/IL-1R) signaling is mediated by Myeloid Differentiation Factor 88 (MYD88), a crucial adapter that mediates the onset of the innate immune response and the generation of proinflammatory cytokines that inhibit pathogens and activate adaptive immunity. While a host's ability to fend off pathogenic infection depends on MYD88, disruption of its abundance can result in autoimmune disorders. One important mechanism for stopping the production of cytokines is the degradation of MYD88.
The AlphaLISA SureFire Ultra Human Total MYD88 Detection Kit is a sandwich immunoassay for the quantitative detection of total MYD88 in cellular lysates, using Alpha technology.
Formats:
- The HV (high volume) kit contains reagents to run 100 wells in 96-well format, using a 60 μL reaction volume.
- The 500-point kit contains enough reagents to run 500 wells in 384-well format, using a 20 μL reaction volume.
- The 10,000-point kit contains enough reagents to run 10,000 wells in 384-well format, using a 20 μL reaction volume.
- The 50,000-point kit contains enough reagents to run 50,000 wells in 384-well format, using a 20 μL reaction volume.
AlphaLISA SureFire Ultra kits are compatible with:
- Cell and tissue lysates
- Antibody modulators
- Biotherapeutic antibodies
Alpha SureFire kits can be used for:
- Cellular kinase assays
- Receptor activation studies
- High-throughput drug screening
How it works
Total-AlphaLISA SureFire Ultra assay principle
The Total-AlphaLISA SureFire Ultra assay measures the expression level of a protein target in a cell lysate.
The Total-AlphaLISA SureFire Ultra assay uses two antibodies which recognize two different distal epitopes on the targeted protein. AlphaLISA assays require two bead types: Acceptor and Donor beads. Acceptor beads are coated with a proprietary CaptSure™ agent to specifically immobilize the assay specific antibody, labeled with a CaptSure tag. Donor beads are coated with streptavidin to capture one of the detection antibodies, which is biotinylated. In the presence of targeted protein, the two antibodies bring the Donor and Acceptor beads in close proximity whereby the singlet oxygen transfers energy to excite the Acceptor bead, allowing the generation of a luminescent Alpha signal. The amount of light emission is directly proportional to the quantity of protein present in the sample.

Total-AlphaLISA SureFire Ultra two-plate assay protocol
The two-plate protocol involves culturing and treating the cells in a 96-well plate before lysis, then transferring lysates into a 384-well OptiPlate™ plate before the addition of Total-AlphaLISA SureFire Ultra detection reagents. This protocol permits the cells viability and confluence to be monitored. In addition, lysates from a single well can be used to measure multiple targets.
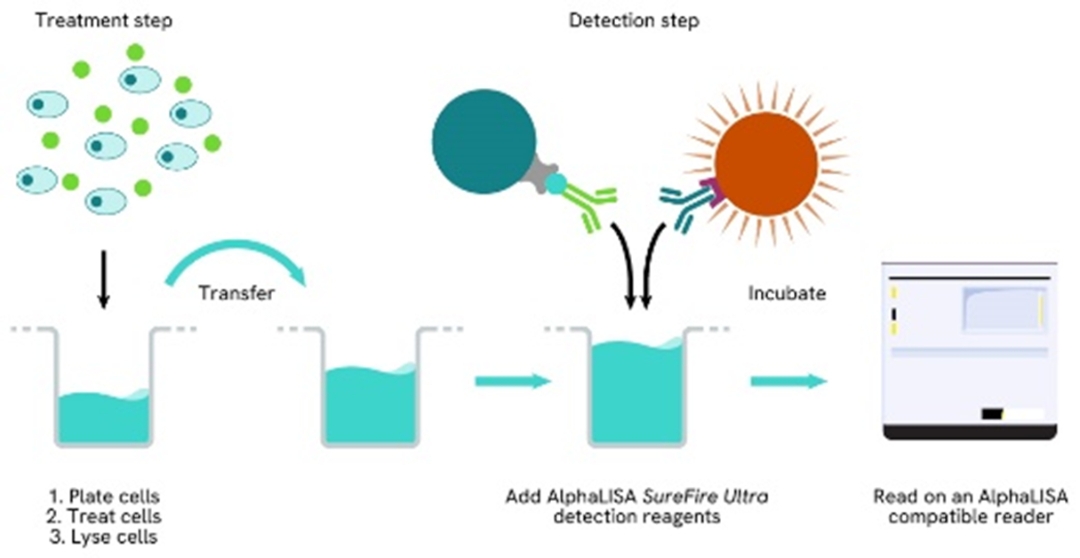
Total-AlphaLISA SureFire Ultra one-plate assay protocol
Detection of Total target protein with AlphaLISA SureFire Ultra reagents can be performed in a single plate used for culturing, treatment, and lysis. No washing steps are required. This HTS designed protocol allows for miniaturization while maintaining AlphaLISA SureFire Ultra quality.
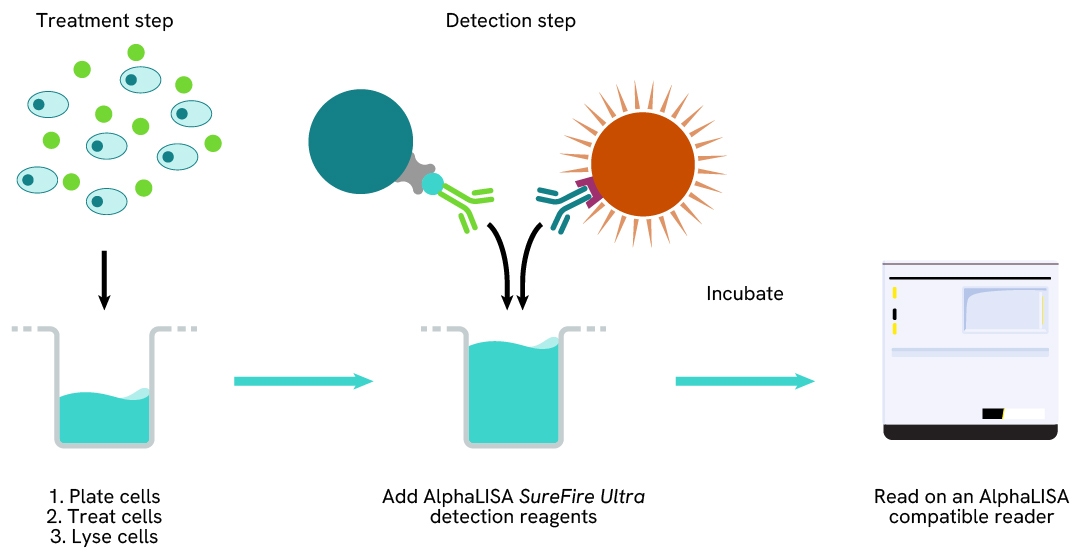
Assay validation
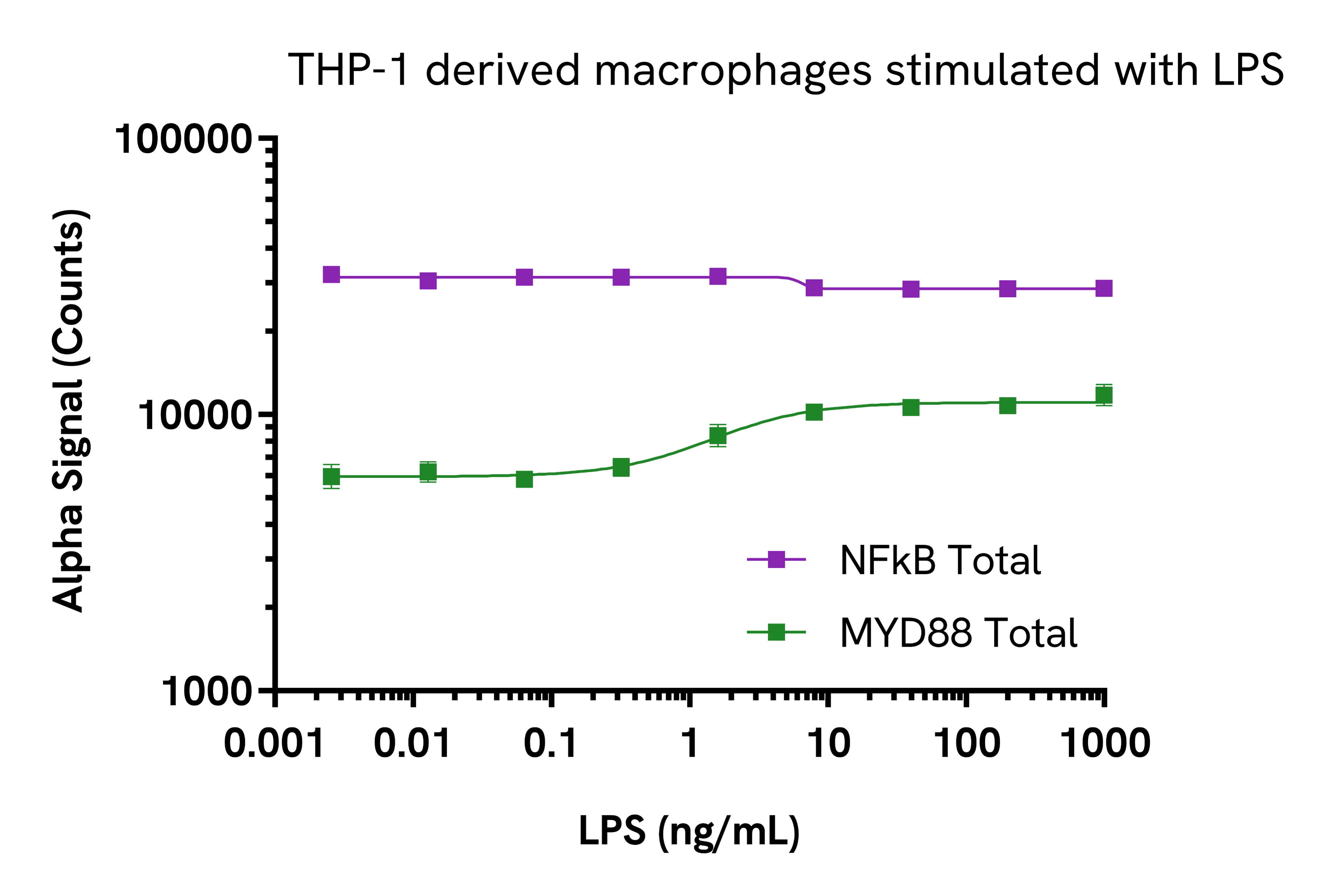
Validation of MYD88 Total assay in THP-1 derived macrophages and Karpas299 cells
THP-1 cells were seeded in a 96-well culture plate (80,000 cells/well) in complete medium, treated with 100 nM PMA and incubated for 24 hours at 37°C, 5% CO2. The cells were further treated with increasing concentrations of LPS for 24 hours. After treatment, the cells were washed in HBSS and lysed with 100 µL of lysis buffer for 10 minutes at RT with shaking at 350 rpm. Lysates were further diluted 1:5 in lysis buffer before proceeding to the detection step. MYD88 Total and NFkB Total levels were evaluated using respective AlphaLISA SureFire Ultra assays. For the detection step, 10 µL of cell lysate (approximately 1,600 cells) were transferred into a 384-well white OptiPlate, followed by 5 µL of Acceptor Mix and incubated for 1 hour at RT. Finally, 5 µL of Donor Mix was then added to each well and incubated for 1 hour at RT in the dark. The plate was read on an Envision Nexus™ using standard AlphaLISA settings.
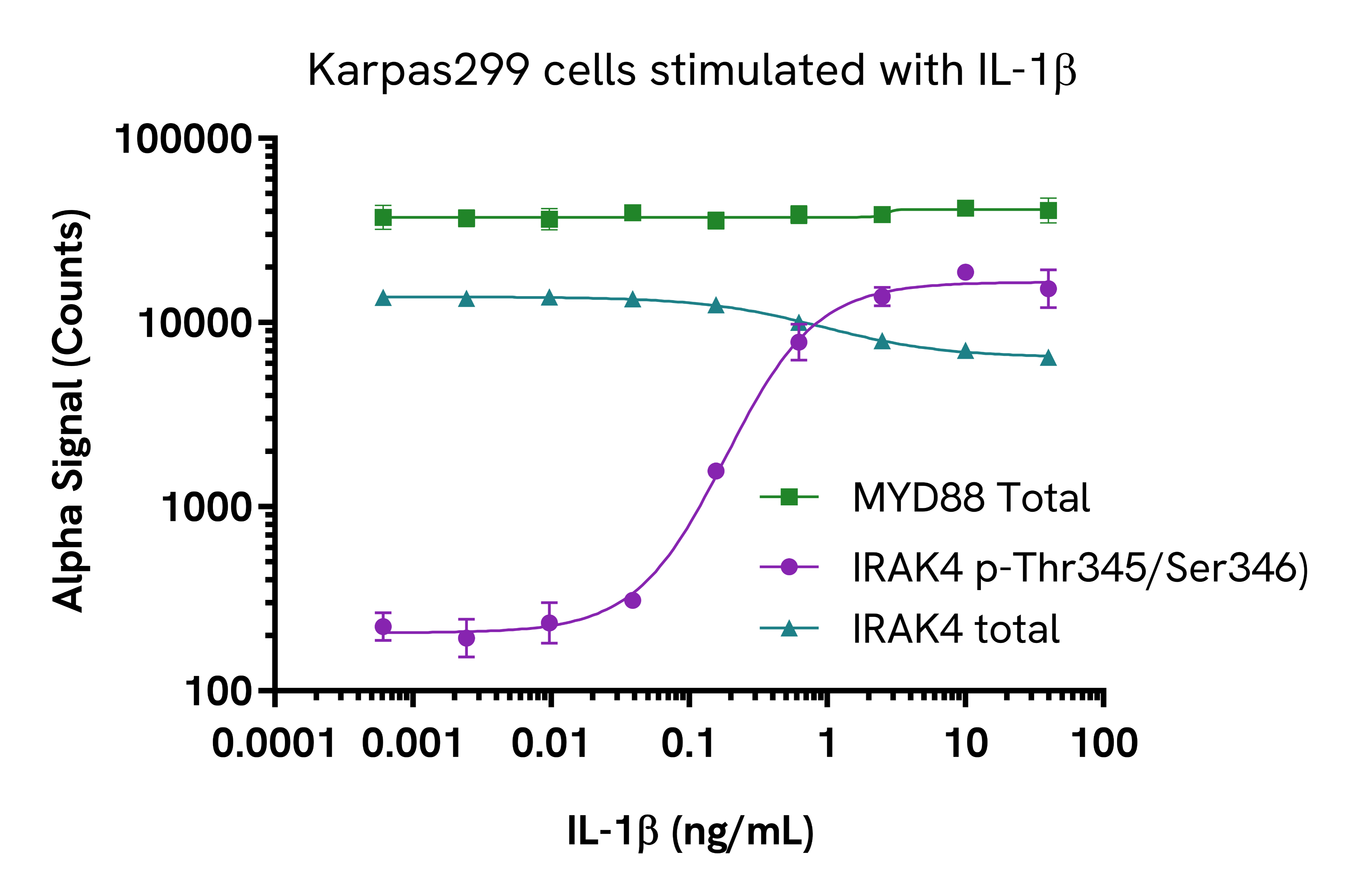
Karpas299 cells were harvested, washed in HBSS + 0.1% BSA and seeded in a 96-well culture plate (100,000 cells/well) in HBSS + 0.1% BSA. The cells were further treated with increasing concentrations of IL-1b for 10 minutes.
After treatment, the cells were lysed with 50 µL of 5X lysis buffer for 10 minutes at RT with shaking at 350 rpm. MYD88 Total, IRAK4 Phospho (Thr345/Ser346) and Total levels were evaluated using respective AlphaLISA SureFire Ultra assays. For the detection step, 10 µL of cell lysate (approximately 4,000 cells) were transferred into a 384-well white OptiPlate, followed by 5 µL of Acceptor Mix and incubated for 1 hour at RT. Finally, 5 µL of Donor Mix was then added to each well and incubated for 1 hour at RT in the dark. The plate was read on an Envision Nexus using standard AlphaLISA settings.
Assay specificity/selectivity
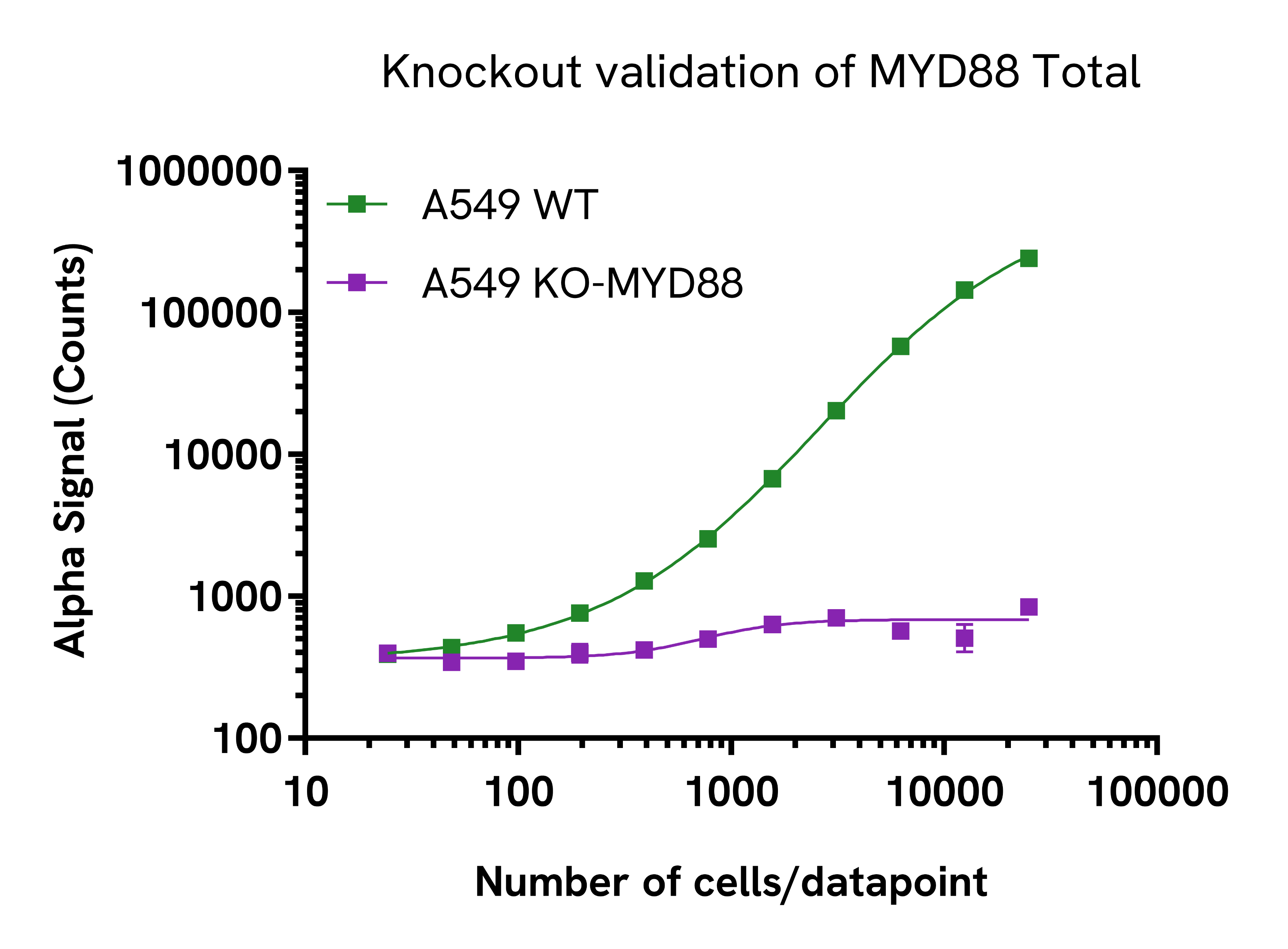
Specificity of MYD88 Total assay
Total MYD88 protein levels were assessed in A549 wild type (WT) cells and MYD88 knockout (KO) A549 cells. Both cell lines were grown to confluency in a T175 flask in complete medium. Each flask was lysed in 4 mL of lysis buffer for 10 minutes at RT with shaking at 350 rpm. Lysates were serially diluted in lysis buffer and evaluated for MYD88 Total as well as Cofilin Total levels to ensure normalization (data not shown). For the detection step, 10 µL of cell lysate were transferred into a 384-well white OptiPlate, followed by 5 µL of Acceptor Mix and incubated for 1 hour at RT. Finally, 5 µL of Donor Mix was then added to each well and incubated for 1 hour at RT in the dark. The plate was read on an Envision Nexus using standard AlphaLISA settings. As expected, MYD88 was detected in the WT-A549 cells but not in the MYD88-KO cell line.
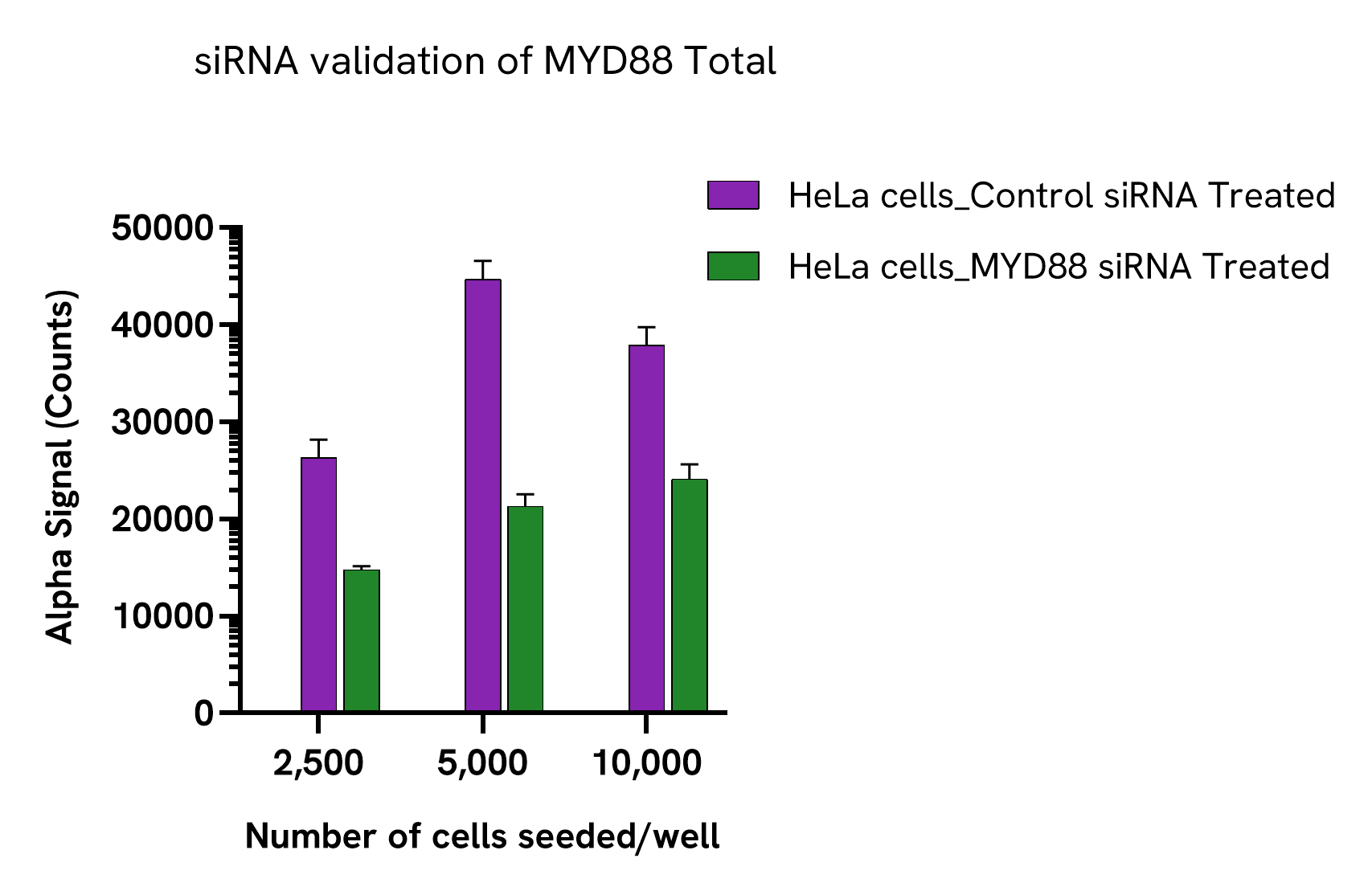
HeLa cells were seeded at different densities in a 96-well plate (200 µL/well) in complete culture medium and incubated overnight at 37°C, 5% CO2. Cells were siRNA treated with 50 µL of a mix of transfection reagent (Santa Cruz, sc-29528)/MYD88 siRNA (Santa Cruz, sc- 35986) or a mix of transfection reagent/siRNA control (Santa Cruz, sc-37007) for 48 hours. After treatment, the cells were lysed with 100 µL of lysis buffer for 10 minutes at RT with shaking at 350 rpm. MYD88 Total levels were evaluated by AlphaLISA SureFire Ultra assays. For the detection step, 10 µL of cell lysate were transferred into a 384-well white OptiPlate, followed by 5 µL of Acceptor Mix and incubated for 1 hour at RT. Finally, 5 µL of Donor Mix was then added to each well and incubated for 1 hour at RT in the dark. The plate was read on an Envision Nexus using standard AlphaLISA settings.
Specifications
| Application |
Cell Signaling
|
|---|---|
| Automation Compatible |
Yes
|
| Brand |
AlphaLISA SureFire Ultra
|
| Cellular or Signaling Pathway |
Inflammasome/Pattern Recognition Receptors (PRRs)
|
| Detection Modality |
Alpha
|
| Host Species |
Human
|
| Lysis Buffer Compatibility |
Lysis Buffer
|
| Molecular Modification |
Total
|
| Product Group |
Kit
|
| Sample Volume |
10 µL
|
| Shipping Conditions |
Shipped in Blue Ice
|
| Target |
MYD88
|
| Target Class |
Phosphoproteins
|
| Target Species |
Human
|
| Technology |
Alpha
|
| Therapeutic Area |
Autoimmunity
|
| Unit Size |
10,000 assay points
|
Video gallery






Resources
Are you looking for resources, click on the resource type to explore further.
Lysine 27 Dimethylation Detection with AlphaLISA Technology
In this technical note discover how AlphaLISA immunodetection assay...
Quantification of Histone H3 Lysine 9 Di-Methylation Using AlphaLISA Assay
In this technical note discover how The AlphaLISA assay...
Loading...


How can we help you?
We are here to answer your questions.






























