
OZ Telepathy CCHD

Automates the capture of Critical Congenital Heart Disease screening from the pulse oximeter or allows for manual entry of CCHD values. Populates CCHD section of OZ eSP™ child health record. The software provides an automatic algorithm calculation of CCHD results. This tool includes a CCHD screening results report (w/ screening timeliness). Simple reporting to state available with state approval. Required for OZ Connect CCHD.
Loading...
Product information
Overview
Congenital Heart Disease is the most common and deadly of all birth defects. Approximately 25 percent of these cases are considered to be “Critical” Congenital Heart Disease (CCHD), particularly hard to detect in the newborn period. CCHD screening has also been proven to be important in identifying secondary conditions (e.g.neonatal sepsis, persistent pulmonary hypertension, parenchymal or anatomic pulmonary disease, transient tachypnea of the newborn, hypothermia, and hemoglobinopathies). These secondary conditions can be detrimental to the newborn if not diagnosed and treated. OZ Telepathy™ CCHD screening solution provides the utmost accuracy in CCHD screening with clinical decision support at the time of screening.
OZ Telepathy CCHD:
- Developed with renowned nursing experts in CCHD screening from Children’s National Medical Center, Heart Institute
- Improve the outcome for newborns when a timely and accurate diagnosis is critical
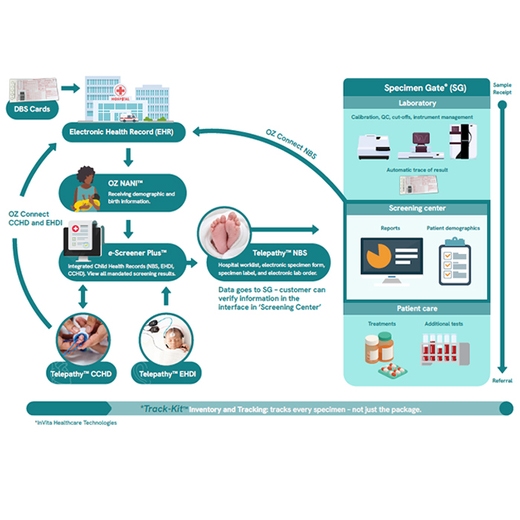
- Provide messaging between a hospital’s electronic health record and OZ software solutions using OZ Newborn Admission Notification Information (NANI™)
- Capture multiple screening values from the pulse oximeter directly into the OZ Telepathy CCHD
- Provide direct clinical decision support to ensure accurate results based on your selected protocol
- Transfer the screening results into the OZ eSP - an integrated child health database - and on to your hospital EHR, other providers or directly to state public health programs
- Provide nurse sensitive quality indicators to monitor program success
- Allow for the monitoring outcomes of the clinical decision support algorithm chosen by your state
Specifications
| Application |
Newborn Screening
|
|---|---|
| Brand |
OZ
|
| Product Group |
Newborn Screening
|
| Research Areas |
Newborn screening
|
| Unit Size |
1 Each
|
Resources
Are you looking for resources, click on the resource type to explore further.
Loading...


How can we help you?
We are here to answer your questions.






























